In the face of escalating global temperatures, understanding how plants perceive and adapt to mild warm conditions is more critical than ever. Recent research illuminates complex molecular mechanisms in Arabidopsis thaliana, revealing how two E3 ubiquitin ligases, MIEL1 and XBAT31, intricately cooperate to regulate thermoresponsive growth. This discovery provides novel insights into plant adaptive responses to ambient temperature increases and opens new avenues for developing heat-resilient crops.
Plant growth and development are profoundly influenced by changes in environmental temperature. Mildly elevated temperatures trigger a phenomenon called thermomorphogenesis, characterized notably by elongation of the hypocotyl—the embryonic stem in seedlings. This elongation facilitates enhanced heat dissipation and survival. Central to this thermoresponsive growth is the transcription factor PHYTOCHROME-INTERACTING FACTOR 4 (PIF4), which integrates temperature signals transmitted by thermosensors such as EARLY FLOWERING 3 (ELF3), phytochrome B (phyB), and PIF7. PIF4 subsequently activates auxin-related gene expression leading to morphological adaptations.
Despite extensive characterization of downstream signaling components, the upstream regulators that finetune temperature perception and signal transmission remain less understood. Prior work has established the involvement of the E3 ubiquitin ligase XBAT31 in promoting thermomorphogenesis by targeting the repressor ELF3 for degradation. By ubiquitinating ELF3, XBAT31 alleviates inhibitory effects on PIF4, permitting enhanced transcriptional activation of heat-responsive genes.
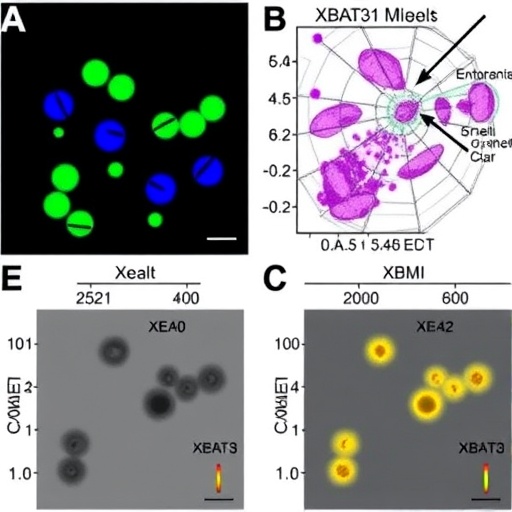
The new study delves deeper into the regulatory landscape by identifying a second E3 ligase, MIEL1, uncovered through yeast two-hybrid screening. Interestingly, MIEL1 exhibits the ability to physically interact with both the full-length XBAT31 protein and its N-terminal domain, a finding confirmed by both in vitro biochemical assays and in vivo experiments. This interaction implies a previously unrecognized layer of control within the thermomorphogenic signaling network.
Phenotypic assessments of Arabidopsis mutants elucidate MIEL1’s positive role in facilitating thermoresponsive hypocotyl elongation. Loss-of-function mutations in MIEL1 significantly reduce hypocotyl growth under mild high-temperature conditions, indicating its essential function upstream of ELF3. Additionally, MIEL1’s genetic positioning relative to XBAT31 implies a hierarchical interaction wherein MIEL1 stabilizes XBAT31 protein abundance.
Explorations into mutual regulatory interactions between MIEL1 and XBAT31 reveal that mutation of MIEL1 leads to a notable decrease in XBAT31 protein levels. Contrarily, disruption of XBAT31 does not impact MIEL1 protein stability. This lack of reciprocal degradation was further supported by in vitro ubiquitination assays, demonstrating that neither ligase ubiquitinates the other, thereby suggesting an indirect mechanism by which MIEL1 influences XBAT31 protein stability.
Further mechanistic insights reveal that XBAT31 molecules engage in self-interaction, a process that facilitates auto-ubiquitination and self-degradation, particularly of the N-terminal domain. Researchers identified eight lysine residues acting as primary auto-ubiquitination sites essential for this degradation pathway. Mutation of these critical ubiquitination sites stabilizes XBAT31N protein, even in the absence of MIEL1, confirming their pivotal role. This auto-catalytic degradation acts as a regulatory checkpoint in controlling XBAT31 protein levels.
Intriguingly, ubiquitination assays show heightened levels of XBAT31N ubiquitination in MIEL1 mutant backgrounds, implicating MIEL1 as a suppressor of XBAT31 auto-ubiquitination. Structural modeling and yeast three-hybrid assays bolster this hypothesis, demonstrating that MIEL1 effectively impedes XBAT31 self-interaction. This inhibition suppresses XBAT31 auto-ubiquitination activity, thereby promoting XBAT31 stabilization and sustained function.
The interplay between MIEL1 and XBAT31 ensures a robust regulatory circuit maintaining optimal XBAT31 levels required for timely degradation of ELF3. This reduction in ELF3 alleviates repression on PIF4, allowing transcriptional activation of auxin-responsive and other heat-adaptive genes fundamental for hypocotyl elongation. Conversely, in MIEL1-deficient plants, increased XBAT31 auto-ubiquitination leads to its degradation, resulting in ELF3 accumulation, dampened PIF4 activity, and suppressed thermomorphogenic growth.
This research uncovers a novel hierarchical cooperation between E3 ubiquitin ligases in plants, where MIEL1 acts as a guardian of XBAT31 protein stability, offering intricate control over thermoresponsive developmental processes. Such detailed molecular understanding sheds light on the sophisticated regulatory networks plants employ to perceive and adapt to environmental fluctuations.
The identification of this MIEL1-XBAT31-ELF3-PIF4 axis not only enhances our comprehension of thermomorphogenesis but also holds significant implications for agriculture amid global warming. By targeting these regulatory components, crop biotechnology can advance strategies to engineer heat-tolerant plant varieties with improved developmental plasticity and resilience.
In conclusion, the interplay between MIEL1 and XBAT31 expands the paradigm of E3 ligase functions beyond substrate ubiquitination to include modulation of other E3 ligases’ stability through suppression of auto-ubiquitination. This study exemplifies the complexity of plant adaptive responses at the proteostatic level and provides promising genetic targets for future breeding programs aimed at mitigating the impacts of rising global temperatures on crop productivity.
Subject of Research: Regulation of thermoresponsive growth via E3 ubiquitin ligases MIEL1 and XBAT31 in Arabidopsis thaliana
Article Title: [Not provided]
News Publication Date: [Not provided]
Web References: http://dx.doi.org/10.1016/j.scib.2026.02.013
Image Credits: ©Science China Press
Keywords: thermomorphogenesis, E3 ubiquitin ligase, MIEL1, XBAT31, Arabidopsis thaliana, ubiquitination, ELF3, PIF4, protein stability, heat stress adaptation, plant development, global warming