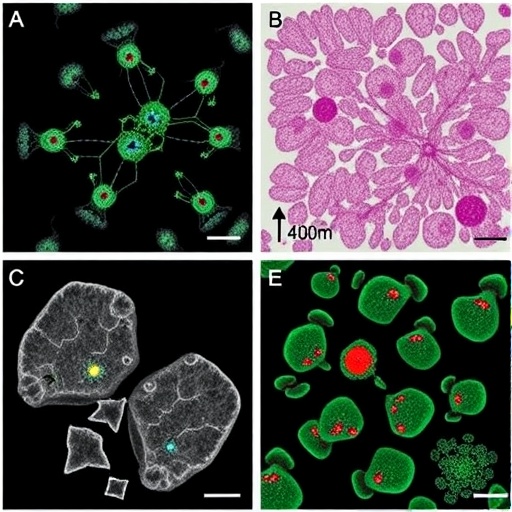
In a groundbreaking advance poised to reshape breast cancer research and mammary gland biology, scientists have unveiled a novel long-term culture system for mouse mammary organoids that faithfully models the intricate physiology of the mammary gland. This cutting-edge platform, coined ‘mini-glands,’ marks a transformative leap in the ability to replicate the dynamic cellular and morphological characteristics of mammary tissue in vitro, bridging critical gaps that have long limited the use of organoid technologies in this field.
Mammary glands are central to a vast spectrum of biological studies, not least due to their role as a key model system for understanding stem cell behavior and breast cancer, which remains the most pervasive female malignancy globally. Traditional two-dimensional cultures and previous organoid systems have struggled to simulate the complex branching architectures and hormonal cycles inherent to the gland’s function. The new mini-gland system remedies these shortcomings by enabling sustained, three-dimensional growth from single basal stem cells into structured organoids displaying remarkable physiological fidelity.
The innovation hinges on a meticulously optimized sequence of growth phases that mirror key developmental and functional milestones observed in vivo. Starting from isolated basal stem cells, the protocol guides cultures through stages of sphere formation, polarity establishment, symmetry breaking, and branching morphogenesis. This progression is carefully choreographed to replicate the natural developmental trajectory of mammary tissue, culminating in organoids with branched architectures analogous to those found in mature glands.
A particularly striking feature of this system is its ability to simulate the mammary gland’s pseudoestrous cycle, encompassing phases analogous to puberty, pregnancy, lactation, and involution. This dynamic replicative capacity allows researchers to observe and manipulate key physiological transitions within a controlled environment. Such functionality is unprecedented in existing mammary organoid models and opens new avenues for studying hormonal regulation and tissue remodeling across reproductive cycles.
At the heart of this protocol is the use of basal stem cells, a critical mammary epithelial population known for its regenerative potential. These cells initiate the developmental cascade, forming spheres that subsequently undergo a series of morphogenic processes driven by precise modulation of culture conditions and extracellular matrix components. The resulting ‘mini-glands’ do not merely mimic static glandular structures but embody the functional complexity and responsiveness of in vivo tissue.
Beyond structural fidelity, the mini-gland culture system is designed to facilitate powerful experimental manipulations. Through genetic tools integrated into the protocol, scientists can perform lineage tracing to track cell fate decisions during organoid maturation. This capability enables comprehensive analysis of stem cell hierarchies, differentiation pathways, and cellular plasticity within the mammary epithelium, providing a window into developmental biology at unmatched resolution.
In parallel, the platform serves as a versatile model for oncogenic transformation studies. By introducing defined genetic alterations into the basal stem cells before organoid formation, researchers can simulate stages of tumor initiation and progression in a physiologically relevant three-dimensional context. This opens the door for high-throughput screening of cancer drivers, gene-environment interactions, and therapeutic responses that have until now been difficult to recapitulate in vitro.
One of the most compelling aspects of the mini-gland system is its accessibility. The comprehensive protocol requires only standard mammalian cell culture expertise and does not depend on specialized instrumentation, lowering the barrier for widespread adoption across research laboratories. This democratization of advanced organoid technology is expected to accelerate discoveries in mammary biology and breast cancer substantially.
The timeline for generating mature, highly branched mini-glands spans approximately two weeks, a period that balances rapid experimental turnaround with faithful morphogenetic progression. Such efficiency contrasts favorably against traditional animal models where studying comparable phenomena can be time-consuming, costly, and ethically complex. The ability to monitor developmental cycles in a dish enhances experimental flexibility and repeatability.
Researchers emphasize that the mini-glands faithfully recapitulate not only gross morphological traits but also the functional aspects of mammary glands, including the cyclic remodeling that underpins reproductive physiology. This dynamic behavior distinguishes the system from previously described organoids, which often represent static or oversimplified tissue states incapable of modeling hormonal environments or physiological flux.
Moreover, the system opens avenues for integrative multi-omics analyses, including transcriptomics, proteomics, and epigenetics, through the precisely controlled in vitro environment. By enabling longitudinal sampling across developmental stages, the mini-glands offer a powerful platform to dissect the molecular underpinnings of normal mammary gland function and disease progression.
This development promises to transform drug discovery pipelines as well, providing a physiologically relevant and scalable model for screening candidate therapeutics targeted toward breast cancer and other mammary gland disorders. The functional and morphological fidelity of the mini-glands heightens the predictive power of preclinical tests, potentially reducing reliance on animal models and enhancing translational success.
As the mammary gland serves as a prototype for branching morphogenesis and epithelial tissue remodeling, the insights gleaned from mini-gland studies may extend beyond breast biology. The principles and protocols devised here could inspire analogous organoid systems for other complex branched organs, amplifying the impact of this work across regenerative medicine and developmental biology.
Looking forward, the scientific community anticipates broad applications of this mini-gland platform, from fundamental studies of mammary stem cell biology to high-throughput genetic screens for cancer susceptibility factors. The ability to capture the full gamut of developmental and pathological stages in vitro heralds a new era of mechanistic understanding and therapeutic innovation for breast cancer and related diseases.
In sum, the establishment of this long-term, versatile culture system represents a landmark achievement in organoid technology, delivering an unprecedented tool that emulates the full architectural and functional complexity of the mammary gland. By harnessing the regenerative power of basal stem cells and meticulously steering morphogenesis, the mini-gland model stands to revolutionize experimental approaches and accelerate breakthroughs in breast cancer research.
Subject of Research:
Development and functional characterization of a long-term culture system for mouse mammary organoids that recapitulates in vivo morphogenesis and physiological cycles, enabling studies in mammary gland biology, stem cell dynamics, and oncogenic transformation.
Article Title:
Establishing a long-term versatile culture system for functional mouse mammary organoids from single cells
Article References:
Yuan, L., Tang, R., Xie, S. et al. Establishing a long-term versatile culture system for functional mouse mammary organoids from single cells. Nat Protoc (2026). https://doi.org/10.1038/s41596-026-01333-1