In a groundbreaking discovery that could redefine our understanding of metabolic regulation and energy homeostasis, researchers have identified a crucial biochemical pathway by which the enzyme AUH, traditionally recognized for its role in leucine catabolism, orchestrates brown adipose tissue (BAT) thermogenesis in male mice. This study provides compelling evidence that AUH influences thermogenic processes not merely through metabolic breakdown of leucine but via a sophisticated mechanism involving post-translational modification of the nuclear receptor PPARγ and an underexplored RNA-binding capacity. The findings, recently published in Nature Communications by Jiang et al., open new avenues for targeting energy expenditure pathways that could have profound implications for treating metabolic disorders such as obesity and diabetes.
Brown adipose tissue, unlike its white counterpart, is specialized for generating heat through non-shivering thermogenesis, a process critical for maintaining body temperature and metabolic health. Central to this function is the nuclear receptor peroxisome proliferator-activated receptor gamma (PPARγ), a transcription factor extensively studied for its role in adipocyte differentiation and lipid metabolism. However, this new research highlights an unexpected regulatory layer wherein PPARγ’s activity is modulated by a novel post-translational modification, specifically HMGylation—a modification involving the addition of a hydroxy-methylglutaryl group—which has not been previously linked to BAT physiology.
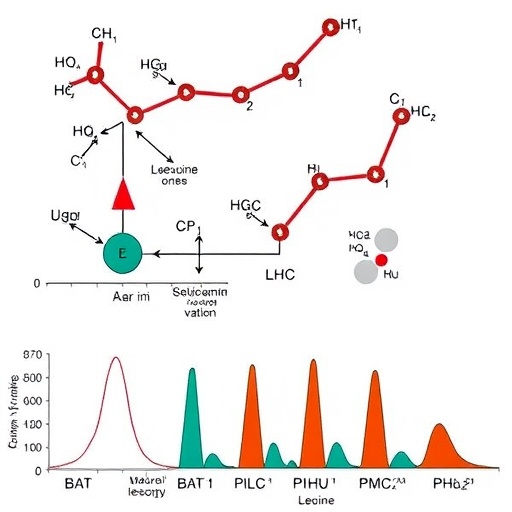
At the heart of this mechanism is AUH (AU RNA-binding protein/enoyl-CoA hydratase), an enzyme traditionally appreciated for catalyzing a key step in leucine catabolism. The enzyme’s ability to catalyze the conversion of methylglutaconyl-CoA to 3-hydroxy-3-methylglutaryl-CoA, a vital intermediate in leucine metabolism, was well documented, yet this study reveals a bifunctional role. Besides enzymatic catalysis, AUH exerts a RNA-binding function that appears to modulate thermogenic gene expression at a post-transcriptional level, suggesting that AUH acts as a metabolic sensor linking amino acid catabolism to thermogenic control.
The researchers utilized a combination of advanced proteomics, transcriptomics, and metabolic phenotyping to demonstrate that in male mice, loss of AUH dampens BAT thermogenesis and decreases energy expenditure, leading to increased adiposity and impaired glucose homeostasis. Mechanistically, AUH facilitates the HMGylation of PPARγ, which augments its transcriptional activity, thereby enhancing the expression of thermogenic genes such as Ucp1. This post-translational modification represents a previously unrecognized mode of fine-tuning PPARγ function in the context of energy metabolism.
Moreover, the RNA-binding aspect of AUH adds a new dimension to thermogenic regulation. The study shows that AUH interacts with specific RNA transcripts in BAT, which likely influences their stability and translation. This dual enzymatic and RNA-binding capability allows AUH to coordinate metabolic inputs from leucine catabolism with gene networks responsible for heat production, effectively coupling nutrient status with energy expenditure.
Another striking element of the study is the sex-specific nature of AUH’s function. The authors report that the regulatory axis involving AUH, PPARγ HMGylation, and RNA-binding predominantly impacts male mice, underscoring complex sexual dimorphisms in BAT biology. This raises exciting questions about how metabolic pathways diverge between sexes and suggests that AUH-targeted therapies might require sex-specific considerations.
This discovery could radically enhance our understanding of the multifaceted control of energy balance, as BAT has been recognized as a promising target for combating metabolic diseases due to its ability to dissipate excess calories as heat. By revealing that an amino acid catabolic enzyme interacts integratively with nuclear receptor signaling and RNA biology, the study broadens the scope of metabolic regulation beyond classical pathways, suggesting new therapeutic targets that capitalize on multifunctional protein enzymes like AUH.
The methodology employed by Jiang and colleagues is equally noteworthy. They combined genetic manipulation techniques, including BAT-specific AUH knockout and overexpression models, with sophisticated mass spectrometry assays capable of detecting PTMs like HMGylation in situ. This allowed for precise mapping of modification sites on PPARγ and assessment of the functional consequences on transcriptional activity. Concurrent RNA immunoprecipitation and sequencing helped delineate AUH’s RNA interaction partners, unveiling a complex post-transcriptional regulatory network.
In addition to the molecular and cellular insights, the physiological assessments confirmed the systemic impacts of AUH modulation. Male mice deficient in AUH exhibited reduced cold tolerance and diminished whole-body energy expenditure, aligning molecular observations with organismal phenotypes. These systemic manifestations underscore the enzyme’s critical role in maintaining metabolic health.
The link between leucine metabolism and thermogenic regulation through AUH also suggests an intriguing metabolic feedback loop. Leucine, a branched-chain amino acid, is an essential nutrient with known effects on mTOR signaling and metabolic health. The coupling of its catabolism to BAT function via AUH implies that dietary and metabolic states could directly influence thermogenic capacity, positioning AUH as a metabolic rheostat that senses nutrient flux and calibrates energy dissipation accordingly.
Furthermore, this research prompts a reevaluation of the functional repertoire of PTMs in nuclear receptor biology. While phosphorylation, acetylation, and ubiquitination of PPARγ have been extensively studied, the identification of HMGylation introduces a new biochemical layer that may have broader implications across different nuclear receptors and transcription factors with pivotal roles in metabolic control.
From a translational perspective, the multifaceted role of AUH holds promise for innovative intervention strategies. Targeting the enzymatic activity or the RNA-binding function of AUH could selectively modulate BAT thermogenesis, offering novel approaches to enhance energy expenditure without the side effects associated with global PPARγ agonists traditionally used in diabetes treatment.
Moreover, the sex-specific findings highlight the importance of personalized medicine in metabolic disease management. Future studies could investigate whether variations in AUH activity contribute to sex-related differences in metabolic disease prevalence and response to therapy, potentially leading to tailored treatments for men and women.
The data also invite closer scrutiny into the role of RNA-binding proteins in metabolism, an emerging field bridging RNA biology with metabolic regulation. AUH exemplifies how multi-domain proteins integrate metabolic cues with gene regulation, potentially inspiring the search for similar multifunctional enzymes in other metabolic tissues.
As exciting as these findings are, the research community must now explore the detailed molecular mechanisms governing AUH’s dual functions and their integration under physiological and pathological states. Questions remain about how HMGylation is dynamically regulated, the spectrum of RNA targets for AUH in BAT and perhaps other tissues, and whether similar mechanisms exist in humans.
In conclusion, the discovery that AUH regulates brown adipose tissue thermogenesis via PPARγ HMGylation and RNA-binding function represents a significant stride forward in metabolic research. It illuminates a nuanced biochemical nexus linking amino acid catabolism to energy expenditure, expanding our grasp of how the body maintains its thermal and metabolic balance. This integrative mechanism not only enhances fundamental understanding but also lays the groundwork for novel therapeutic strategies targeting metabolic diseases through modulation of multifunctional enzymes like AUH.
Subject of Research: Regulation of brown adipose tissue thermogenesis by the leucine catabolic enzyme AUH through PPARγ HMGylation and RNA-binding in male mice.
Article Title: Leucine catabolic enzyme AUH regulates BAT thermogenesis via PPARγ HMGylation and RNA-binding function in male mice.
Article References: Jiang, H., Ni, S., Li, Z. et al. Leucine catabolic enzyme AUH regulates BAT thermogenesis via PPARγ HMGylation and RNA-binding function in male mice. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71581-y
Image Credits: AI Generated