In a groundbreaking breakthrough that promises to redefine our understanding of muscle physiology, researchers have uncovered a pivotal regulatory mechanism rooted deep within the cell’s lysosomes, fundamentally linking lipid signaling to muscle growth. The new study, published in Nature Metabolism, reveals how lysosomal phosphoinositide turnover serves as a crucial upstream modulator of the Rag GTPase-mTORC1 signaling axis, consequently orchestrating muscle mass expansion. This discovery not only opens new vistas in muscle biology but also unveils promising therapeutic avenues for muscle-wasting conditions and metabolic disorders.
The mammalian target of rapamycin complex 1 (mTORC1) is well-established as a master regulator of cellular growth and metabolism, responding dynamically to nutrient availability, energy status, and growth signals. Central to this process is the Rag GTPase complex, which governs mTORC1 localization to lysosomal membranes where it becomes activated. Until now, the complexity of signaling events upstream to Rag GTPases and how lysosomal lipid dynamics influence this pathway remained poorly understood. The team led by Picot et al. bridges this gap by demonstrating that the turnover of specific phosphoinositides within lysosomal membranes dictates Rag GTPase activity, acting as a molecular rheostat for mTORC1 signaling and muscle development.
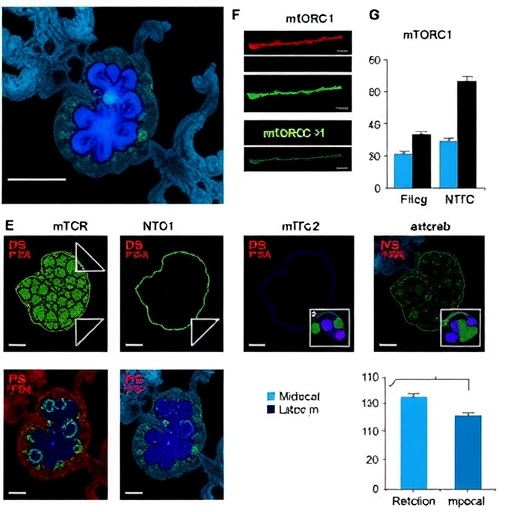
At the heart of these findings lies phosphoinositides—specialized phosphorylated lipids that regulate diverse cellular processes by virtue of their spatial and temporal distribution on membranes. Lysosomal phosphoinositides, in particular, have been enigmatic given the organelle’s traditional classification as the cellular degradation hub. The study overturns this view, showcasing lysosomes as dynamic signaling platforms where rapid phosphoinositide turnover finely tunes muscle cellular anabolic pathways. Utilizing advanced lipidomic analyses coupled with sophisticated genetic perturbations in murine models, researchers pinpoint dynamic changes in phosphatidylinositol 3-phosphate (PI3P) and phosphatidylinositol 3,5-bisphosphate (PI(3,5)P2) levels as decisive factors modulating Rag GTPase localization and activity.
Mechanistically, the research unravels an intricate feedback loop where phosphoinositide-modifying enzymes coordinate lysosomal membrane lipid composition. These changes alter binding affinities of Rag GTPase regulators, thereby finely adjusting mTORC1 recruitment and activation. Disrupting this lipid turnover process uncouples Rag GTPase activity from nutrient cues, resulting in blunted mTORC1 signaling and impaired muscle hypertrophy. Conversely, enhancing lysosomal phosphoinositide cycling boosts mTORC1 output and fosters robust muscle growth, establishing causation beyond correlation.
Excitingly, the study reveals muscle-specific nuances in lysosomal lipid signaling. Unlike other tissues, skeletal muscle fibers exhibited heightened sensitivity to phosphoinositide dynamics, suggesting tissue-specific adaptions that align growth demands with metabolic needs. This insight underscores the lysosome’s broader role as a nutrient-sensing organelle tailored to meet the unique bioenergetic and biosynthetic requirements of muscle physiology. Moreover, these observations may recalibrate current paradigms explaining muscle adaptation during exercise and pathological atrophy.
From a methodological perspective, the team’s integrated approach—combining high-resolution confocal microscopy, lipid mass spectrometry, and CRISPR-Cas9-mediated gene editing—provided an unprecedented window into the spatiotemporal orchestration of lipid turnover and signaling. These techniques illuminated the delicate choreography of lysosomal lipid remodeling happening in real time within intact muscle fibers, an accomplishment that represents a technical tour de force and sets the stage for future mechanistic explorations.
Additionally, the research highlights potential intersections between lysosomal lipid metabolism and other anabolic pathways. The cross-talk between phosphoinositide turnover and autophagic flux, another lysosome-centered process, emerged as a tantalizing area meriting further study. Since autophagy plays a dual role in cell maintenance and remodeling, decoding the interplay between these processes may shed light on muscle plasticity under varying physiological and pathological stresses.
The translational implications are profound. Muscle wasting associated with aging, cachexia, and various chronic diseases remains a significant clinical challenge with limited therapeutic options. The identification of lysosomal phosphoinositide turnover as a critical node upstream of mTORC1 presents novel targets for intervention. Small molecule modulators or gene therapies designed to enhance or restore appropriate lysosomal lipid dynamics could revitalize anabolic signaling pathways and preserve muscle mass and function.
Furthermore, because mTORC1 signaling extends its influence into metabolic regulation, including glucose homeostasis and lipid metabolism, the findings may ripple beyond muscle tissue. Targeting lysosomal lipid turnover might emerge as a multipronged strategy to ameliorate metabolic dysregulations seen in diabetes and obesity. Careful delineation of these systemic effects will be vital.
Importantly, this work challenges the simplistic notion that lysosomes serve merely as end-stage degradation centers. Instead, it aligns with a growing body of evidence positioning lysosomes as versatile signaling hubs where lipid modifications actively regulate cell signaling cascades. This conceptual shift can inspire renewed efforts to characterize lipid dynamics on intracellular membranes across diverse biological contexts.
As muscle strength and mass correlate strongly with healthspan and survival in humans, optimizing muscle anabolic pathways has wide-reaching public health implications. Unlocking the lysosomal lipid code that governs mTORC1 could lead to innovative exercise mimetics or nutritional supplements tailored to augment muscle functionality, particularly in vulnerable populations such as the elderly.
In summary, the work by Picot et al. orchestrates an elegant convergence of cell biology, lipidomics, and muscle physiology to unveil lysosomal phosphoinositide turnover as an indispensable upstream signal for Rag GTPase–mTORC1 activation and muscle growth. It propels lysosomes to center stage as active integrators of nutrient and growth signals, expanding the molecular framework governing muscle anabolism. This landmark discovery not only advances basic scientific knowledge but also charts a promising path toward therapeutic innovation against muscle degeneration.
As the research community digests these insights, future investigations will undoubtedly explore how manipulating lysosomal lipid metabolism could fine-tune mTORC1 outputs across tissues, expand our understanding of lysosomal signaling networks, and ultimately improve muscle health in aging and disease. The marriage of lipid signaling to classical anabolic pathways stands as a testament to the layered complexity of cellular growth control, heralding a new era of metabolic precision medicine.
Subject of Research: Lysosomal phosphoinositide turnover regulation of Rag GTPase–mTORC1 signaling and its impact on muscle growth
Article Title: Lysosomal phosphoinositide turnover acts upstream of RagGTPase–mTORC1 and controls muscle growth
Article References:
Picot, M., Hifdi, N., Vaucourt, M. et al. Lysosomal phosphoinositide turnover acts upstream of RagGTPase–mTORC1 and controls muscle growth. Nat Metab (2026). https://doi.org/10.1038/s42255-026-01484-1
Image Credits: AI Generated
DOI: https://doi.org/10.1038/s42255-026-01484-1