In a groundbreaking advancement that bridges genetics and neuroimaging, a new study has illuminated the subtle yet profound effects of a specific genetic anomaly on brain white matter in mid-to-late adulthood. Researchers have meticulously examined individuals carrying the 15q11.2 copy number variation (CNV), a small but significant deletion or duplication on chromosome 15 long arm region 11.2, revealing how these structural genetic differences manifest in brain microstructure changes compared to non-carriers. Published in Translational Psychiatry, this research propels forward our understanding of how rare genetic variations could influence brain aging and neurological health.
The 15q11.2 CNV, characterized by either the deletion or duplication of a genomic segment encompassing several genes, has been implicated in various neurodevelopmental disorders and psychiatric conditions. However, until now, the impact of this variant on the adult brain’s white matter integrity had not been comprehensively understood. White matter, composed of myelinated nerve fibers, serves as the brain’s communication highway, enabling rapid signal transmission between different regions. Alterations in white matter microstructure are known to underpin cognitive decline and are associated with disorders such as schizophrenia, autism, and dementia.
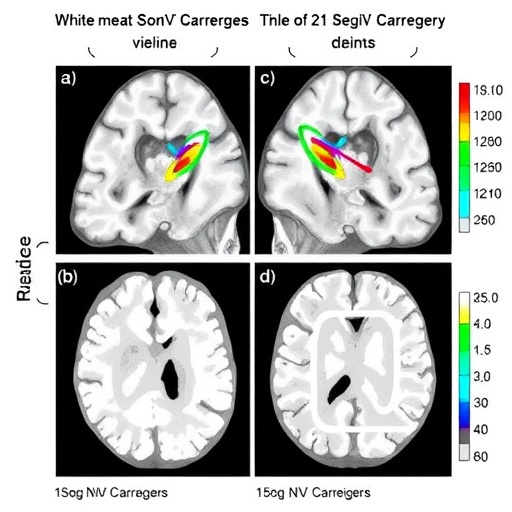
This study employed advanced neuroimaging techniques, including diffusion tensor imaging (DTI), to quantify white matter microstructural properties in a large cohort of mid-to-late life adults stratified by 15q11.2 CNV carrier status. DTI enables measurement of parameters such as fractional anisotropy (FA) and mean diffusivity (MD), which serve as biomarkers for white matter integrity—indicating the coherence and density of fiber tracts. By integrating genetic data with brain imaging from population-based databases, the researchers could isolate subtle yet statistically robust differences associated with the CNV.
Their analysis found that carriers of the 15q11.2 deletion exhibited reduced FA and increased MD in several major white matter tracts relative to non-carriers, suggesting compromised fiber coherence and possible demyelination or axonal loss. These changes were localized particularly in regions such as the corpus callosum, superior longitudinal fasciculus, and internal capsule, all critical pathways for interhemispheric communication and cognitive function. Meanwhile, duplication carriers showed a more complex, region-dependent pattern of deviations, indicating that both loss and gain of genetic material can disrupt brain connectivity, albeit through potentially distinct mechanisms.
Intriguingly, these microstructural differences emerged predominantly in mid-to-late life cohorts, implicating the CNV in influencing brain aging trajectories rather than solely early neurodevelopment. This temporal specificity hints at an age-related vulnerability aggravated by the genetic variation, possibly predisposing carriers to accelerated neurodegeneration or diminished cognitive reserve. Importantly, these findings link genetic risk at the microscopic white matter level to macroscopic brain aging processes, which could have implications for early detection and personalized interventions.
The authors propose that the genes located within the 15q11.2 segment—such as NIPA1, NIPA2, CYFIP1, and TUBGCP5—may influence the expression of oligodendrocytes or myelin maintenance mechanisms. Aberrations in these cellular processes could logistically explain the imaging phenotypes observed. For example, CYFIP1 has been shown to interact with cytoskeletal elements critical for myelin sheath integrity, and disruptions therein may lead to the structural deficits detected by DTI. By bridging gene function with imaging phenotypes, the study opens avenues for mechanistic exploration in cellular and animal models.
Moreover, these white matter alterations may contribute to cognitive and neuropsychiatric outcomes previously associated with 15q11.2 CNVs, including increased susceptibility to psychiatric illnesses like schizophrenia and epilepsy. While this study did not directly assess clinical correlates, it lays a foundation for future longitudinal investigations correlating structural changes with cognitive trajectories and psychiatric symptoms in carriers, fostering a better understanding of genotype-phenotype relationships.
From a methodological perspective, the study leveraged rigorous statistical control for confounding variables including age, sex, and overall brain volume, strengthening the credibility of the associations reported. Additionally, the use of large, community-based cohorts ensured generalizability beyond clinical samples, highlighting the relevance of rare genetic variants in the general aging population’s brain health.
Clinically, these insights could inform biomarker development and risk stratification strategies, enabling clinicians to identify individuals at heightened risk of white matter pathology and cognitive decline due to their genetic background. Interventions, be they pharmacological or lifestyle-oriented, could be tailored to mitigate white matter damage or enhance repair processes in genetically susceptible populations. This personalized medicine approach aligns with the growing emphasis on integrating genomics with neuroimaging and cognitive phenotyping.
The study also raises provocative questions about the complexity of genetic influences on brain aging: how do different CNVs interplay with environmental exposures, vascular risk factors, and other molecular cascades influencing white matter integrity? Do lifestyle factors such as physical exercise or cognitive engagement modulate white matter changes in 15q11.2 CNV carriers? These avenues stand ripe for future multidisciplinary research.
Furthermore, this research exemplifies the potential of combining high-resolution brain imaging with genetic data to uncover biological signatures of aging and disease risk. As computational tools and imaging technologies advance, the granularity with which scientists can characterize brain microstructure will only increase, bringing us closer to precise mappings of genetic variation onto brain architecture and function.
In sum, the discovery of white matter microstructure differences in 15q11.2 CNV carriers represents a landmark step toward unraveling the genetic contributions to brain integrity in aging. With implications spanning fundamental neuroscience to clinical practice, this work not only deepens our understanding of neurogenetics but also charts actionable paths toward personalized neuroprotection.
As this research continues to inspire further studies, it underscores that the architecture of the aging brain is not merely shaped by environmental and lifestyle factors but also deeply by our unique genetic code. The subtle variations encoded in our DNA may set us on trajectories of resilience or vulnerability, ultimately influencing cognitive health and quality of life as we age.
By integrating genomic insights with neuroimaging and clinical data, science is edging closer to deciphering the complex symphony of factors orchestrating brain aging. The 15q11.2 CNV stands out as a compelling genetic player, and understanding its impact could unlock strategies for enhancing brain health in an aging global population.
This pivotal work invites us to rethink the narrative around rare genetic variants—not as isolated anomalies—but as meaningful contributors to the brain’s lifelong structural evolution and potential targets for therapeutic innovation.
Subject of Research: White matter microstructure differences in mid-to-late life individuals carrying 15q11.2 copy number variations.
Article Title: White matter microstructure differences between 15q11.2 copy number variation carriers and non-carriers in mid-to-late life.
Article References: Korbmacher, M., Boen, R., Andreassen, O.A. et al. White matter microstructure differences between 15q11.2 copy number variation carriers and non-carriers in mid-to-late life. Transl Psychiatry (2026). https://doi.org/10.1038/s41398-026-03962-2
Image Credits: AI Generated