In a groundbreaking study that promises to transform our understanding of liver biology and disease, researchers have uncovered a critical cellular mechanism by which the liver maintains its delicate balance of bile acid (BA) regulation and tissue integrity. The study, published in Nature Metabolism, reveals an intricate signalling axis involving the farnesoid-X-receptor (FXR) and Yes-associated protein (YAP) within biliary epithelial cells (BECs), which line the bile ducts and serve as vital guardians of liver homeostasis. Intriguingly, the FXR–YAP pathway acts as a molecular sentinel, preserving the identity and function of BECs, while preventing the pathological fibrosis that marks the progression from liver injury to cirrhosis.
Bile acids, synthesized in hepatocytes, are secreted into the biliary system to aid in lipid digestion. They transit through a network of ducts lined with BECs, whose role extends far beyond simple conduits. These epithelial cells form a tightly regulated barrier that maintains ductal integrity, preventing toxic BA spillover into the surrounding liver parenchyma. Dysfunction in this barrier has long been known to precipitate BA-induced damage, inflammation, and eventually fibrosis—a pathological scarring process that disrupts liver architecture and compromises function. However, the cellular mechanisms underlying how BECs sustain barrier function and restrain fibrogenesis have remained elusive—until now.
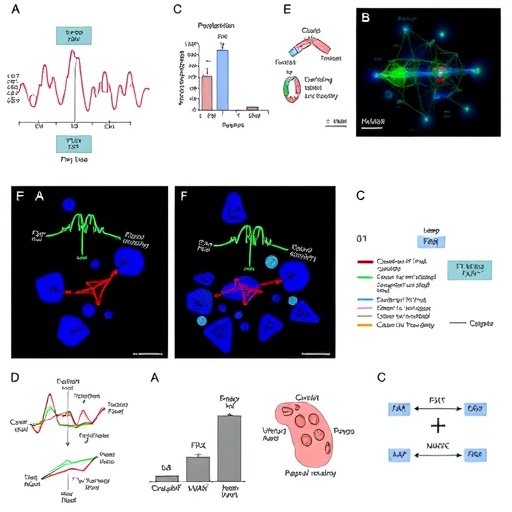
The team employed a sophisticated combination of mouse genetic models, computational gene regulation analyses, and validation with human liver samples to dissect this regulatory axis. They found that FXR, a nuclear receptor traditionally recognized for its role in systemic bile acid sensing and metabolic control, is highly expressed in BECs. Significantly, FXR acts as a transcriptional activator of YAP, a potent regulator of cell proliferation and tissue repair pathways. This FXR-driven activation of YAP enables BECs to maintain robust adhesive properties crucial for the integrity of the biliary barrier, effectively preventing the leakage of bile acids into parenchymal tissue.
Loss-of-function experiments using mouse models with FXR or YAP selectively ablated in BECs uncovered a dramatic cascade of pathological events, highlighting the indispensability of this signalling duo. Without FXR or YAP, BECs undergo profound phenotypic changes characterized by activation of β-catenin signalling and a mesenchymal-like transition. These changes provoke abnormal BEC proliferation and a breakdown of the bile duct barrier, resulting in unchecked bile acid diffusion, hepatic stellate cell activation, and the establishment of a fibrotic microenvironment. This pathological sequence critically accelerates fibrogenesis and pushes liver injury toward irreversible cirrhosis.
An especially compelling aspect of this study is its translational relevance. Human liver biopsies demonstrated a striking correlation between diminished FXR–YAP signalling within BECs and the severity of fibrosis, cementing the clinical significance of these findings. This implies that weakened FXR–YAP pathways could serve as biomarkers for progressive liver disease and potentially predict patient outcomes. Moreover, the group’s clinical insights raise caution against the use of obeticholic acid (OCA), a synthetic FXR agonist currently employed in treating certain cholestatic liver diseases. While OCA typically activates FXR to restore bile acid homeostasis, in the context of FXR-deficient BECs, its administration exacerbated fibrotic progression, uncovering a paradoxical effect that may inform future therapeutic strategies.
Mechanistically, this research highlights how bile acids intrinsically “reprogram” BECs via FXR to engage YAP-dependent transcriptional networks. This dynamic programming preserves epithelial identity, reinforcing junctional adhesion complexes that fortify the ductal wall. Concurrently, FXR–YAP signalling curtails β-catenin-mediated mesenchymal transformation, which otherwise endangers biliary architecture and promotes fibrotic niche formation. This nuanced interplay ensures that BECs not only provide structural support but serve as active regulators of tissue homeostasis, engaging in crosstalk with hepatic stellate cells and the broader liver microenvironment.
The implications of this study extend well beyond the specialized realm of bile duct biology. By positioning FXR–YAP as a central molecular axis in epithelial maintenance and fibrosis prevention, these findings invite deeper exploration into related pathways involved in other epithelial tissues subject to fibrogenic insults. Given the notorious difficulty in treating liver fibrosis once established, unravelling endogenous mechanisms that halt fibrogenesis upstream offers a beacon of hope for more effective, targeted interventions that restore normal tissue architecture rather than merely mitigating symptoms.
Future investigations spurred by these discoveries will likely focus on identifying small molecules or biologics that potentiate the FXR–YAP axis specifically within BECs, potentially offering novel antifibrotic therapies with enhanced precision and safety profiles. Additionally, understanding how BA dysregulation influences BEC signalling in chronic liver diseases such as primary sclerosing cholangitis, non-alcoholic steatohepatitis, and cholangiocarcinoma may unlock new paradigms for managing these conditions.
This study also underscores the remarkable plasticity of epithelial cells in the liver, highlighting their dual capacity to act as both damage responders and guardians of tissue integrity. The observation that epithelial cells translate metabolic cues from bile acids into structural and transcriptional programs reshapes our conceptual framework of liver biology, reinforcing the integrative nature of metabolism, signalling, and cellular identity.
In summary, the elucidation of the FXR–YAP signalling pathway in BECs represents a major advance in hepatology, revealing how these specialized epithelial cells autonomously safeguard the bile duct barrier and modulate the fibrogenic cascade. By intricately balancing adhesion, proliferation, and mesenchymal transition, FXR–YAP ensures that bile acid homeostasis is maintained and fibrotic progression curtailed. The clinical and therapeutic ramifications of this discovery herald a promising frontier for liver disease research, with the potential to pivot current treatment paradigms toward preserving endogenous tissue homeostasis and preventing cirrhosis before its deadly culmination.
Subject of Research: Biliary epithelial cell biology, bile acid regulation, liver fibrosis, FXR–YAP signalling pathways.
Article Title: FXR–YAP signalling maintains biliary epithelial cell identity and preserves liver homeostasis.
Article References:
Sánchez-Sánchez, P., Wang, Z., Zagorac, S. et al. FXR–YAP signalling maintains biliary epithelial cell identity and preserves liver homeostasis. Nat Metab (2026). https://doi.org/10.1038/s42255-026-01521-z
Image Credits: AI Generated