In a groundbreaking study set to redefine the use of widely employed neuronal cell models, researchers have unveiled critical insights into the limitations of SH-SY5Y cells, a staple in neurobiological research. The team, comprised of Leuenberger, Ott, Nevian, and collaborators, has meticulously characterized the neuronal differentiation of SH-SY5Y cells, revealing that while these cells manifest typical neuronal markers and morphological features, they fundamentally lack the complex synaptic maturity necessary to model fully functional neurons in vitro. This distinction holds profound implications for neuroscience research, drug discovery, and understanding neurodegenerative disease mechanisms.
Differentiated SH-SY5Y cells have long been utilized as a convenient, human-derived model to simulate neuron-like properties, allowing researchers to study neuronal behavior without the complexity and ethical challenges of primary human neurons. However, despite their widespread adoption, uncertainties surrounding the extent to which SH-SY5Y cells replicate true neuronal function have persisted. This new study, published in Cell Death Discovery in 2026, systematically deconstructs the cellular and molecular features of differentiated SH-SY5Y cells, highlighting a crucial gap in synaptic development that challenges the validity of this model for synaptic physiology studies.
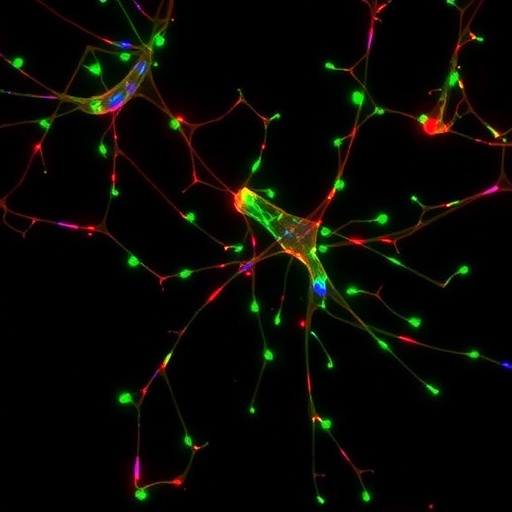
The researchers embarked on a comprehensive evaluation of SH-SY5Y cells differentiated under established protocols, carefully assessing neuronal morphology, cytoskeletal organization, and expression of canonical neuronal markers such as MAP2, βIII-tubulin, and NeuN. Indeed, the cells exhibited pronounced neurite outgrowth consistent with early neuronal differentiation and showed expression profiles aligned with immature neurons. Confocal microscopy confirmed cytoskeletal reorganizations characteristic of neuronal cells; however, when probed for synaptic proteins such as synaptophysin and PSD-95, markers critical for functional synaptic assembly, the results were underwhelming.
Electrophysiological assays further cemented the narrative, with differentiated SH-SY5Y cells failing to display robust synaptic activity or the characteristic synaptic currents observed in mature neurons. Despite visible neurite formation, the lack of spontaneous excitatory or inhibitory postsynaptic currents suggested incomplete synaptogenesis. The absence of mature synaptic machinery indicates that while these cells are useful for modeling neuronal differentiation, they fall short as surrogates for synaptic transmission and plasticity studies.
The implications of these findings are far-reaching. Many studies investigating neurodegenerative disease pathways, neurotoxicity, and synaptic pharmacology have relied heavily on SH-SY5Y cells, often extrapolating findings to mature neuronal function. The revelation that SH-SY5Y cells do not develop synaptic maturity calls for a critical reassessment of past conclusions drawn from this model. Researchers may need to pivot toward more physiologically relevant systems, such as induced pluripotent stem cell (iPSC)-derived neurons or primary neuronal cultures, especially when synaptic activity is central to the investigation.
Furthermore, the study underscores the importance of rigorous validation of in vitro models. The allure of SH-SY5Y cells lies in their ease of culture and human origin, but as this research highlights, morphological and marker expression alone are insufficient to confirm functional neuronal identity. In neurobiology, synaptic integration and neurotransmission form the bedrock of neuronal communication and network function; absence of these properties in the model fundamentally limits translational relevance.
Highlighting another layer of complexity, the investigation discusses the differentiation protocols themselves. Variability in differentiation time, retinoic acid treatment duration, and supplements significantly affect neuronal maturation outcomes. Although the study employed robust and widely accepted protocols, the intrinsic limitations of the SH-SY5Y lineage impose a ceiling on achievable maturity levels. This insight may spur efforts to optimize differentiation paradigms or engineer new cell lines with enhanced capacity for synaptic development.
On a molecular scale, the study dives into gene expression profiles associated with synaptogenesis, signaling pathways, and cytoskeletal dynamics. Transcriptomic analyses revealed downregulation of genes pivotal for synapse formation and synaptic vesicle cycling, further confirming the phenotypic observations. Such molecular signatures establish a blueprint for identifying bottlenecks in SH-SY5Y maturation and could guide genetic or pharmacological interventions aimed at enhancing synaptic features.
From a technical perspective, this research elegantly integrates state-of-the-art imaging techniques, including high-resolution immunofluorescence and live-cell imaging, with electrophysiological analyses such as patch-clamp recordings to deliver a multidimensional view of neuronal characteristics. The multidisciplinary approach provides a compelling and comprehensive profile, reinforcing the robustness of the conclusions.
The broader neuroscience community stands to benefit immensely from these revelations. By clarifying the capabilities and limitations of SH-SY5Y cells, the study equips researchers with vital information to select appropriate models tailored to their scientific questions, ultimately increasing the rigor and reproducibility of neurobiological research.
In addition, the paper encourages the adoption of complementary models that can recapitulate the complexity of synaptic networks. Emerging platforms, including organoid systems and co-culture models, may offer more physiologically relevant environments to investigate synaptic connectivity and disease pathology, bridging the gap left by simplified cell lines.
This research also revitalizes interest in exploring the fundamental biology of synaptic development. By contrasting the differentiated SH-SY5Y cells with mature neurons, the study flags key molecular players and pathways that govern synaptic assembly—a fertile ground for future research aiming to manipulate neuronal maturity therapeutically.
The timing of this publication is particularly impactful given the accelerating demand for human-relevant neurobiological models suitable for high-throughput drug screening and precision medicine. As the field moves toward personalized neural models, ensuring the functional fidelity of in vitro systems cannot be overstated.
Ultimately, this seminal study redefines our understanding of a ubiquitous model system, blending technical sophistication with biological insight. It highlights a critical bottleneck in neuronal modeling and sets the stage for advances that could propel neuroscience, neuropharmacology, and neurology into a new era of accuracy and relevance.
As research endeavors continue to unravel the complexities of neuronal function, the work by Leuenberger and colleagues serves as both a cautionary tale and an inspiration—a reminder that shining a light on model limitations is essential for scientific progress and that the quest for models reflecting true neuronal authenticity remains a vibrant challenge on the horizon.
Subject of Research: Neuronal differentiation and synaptic maturity of SH-SY5Y cells in vitro.
Article Title: Differentiated SH-SY5Y cells exhibit neuronal features but lack synaptic maturity.
Article References:
Leuenberger, J., Ott, G., Nevian, T. et al. Differentiated SH-SY5Y cells exhibit neuronal features but lack synaptic maturity. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-03094-y
Image Credits: AI Generated