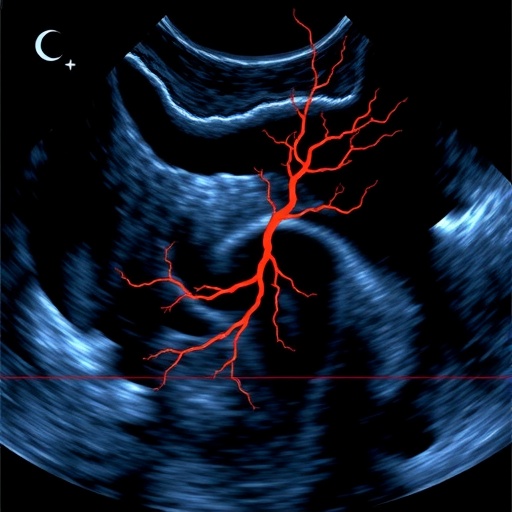
In a revolutionary leap for biomedical imaging, researchers at Mayo Clinic have unveiled a cutting-edge ultrasound technique known as Radon transform-based flow measurement, or R-Flow, heralding a new era in microvascular visualization. Published recently in the journal BME Frontiers, this innovative method transcends the long-standing limitations of conventional Doppler ultrasound, enabling contrast-free, real-time, and angle-independent mapping of blood flow dynamics at the microscopic level. This breakthrough holds transformative potential for diagnosing and monitoring vascular pathologies such as liver cirrhosis, where detailed insights into microcirculation are paramount.
Ultrasound has long been a staple in clinical imaging due to its safety and accessibility, yet traditional Doppler techniques struggle with multidirectional flow assessment and often require contrast agents to enhance visualization. R-Flow radically departs from this paradigm by employing the Radon transform—a sophisticated mathematical tool—to decode red blood cell trajectories directly from ultrafast plane-wave ultrasound signals. This approach allows it to simultaneously reconstruct both axial (along the ultrasound beam) and lateral (perpendicular to the beam) velocity components, delivering comprehensive two-dimensional flow vector maps.
The technical core of R-Flow involves acquiring beamformed in-phase and quadrature (IQ) data using ultrafast plane-wave ultrasound imaging, which samples the microvascular bed at extremely high frame rates. Subsequent clutter filtering isolates the blood flow signals from stationary tissue echoes, enabling the construction of spatiotemporal image slices along lateral-time (x–t) and axial-time (z–t) planes. By applying the Radon transform to these slices, researchers extract directionally resolved velocity components with exceptional precision. Additionally, axial motion compensation is applied during lateral flow estimation to enhance the continuity of red blood cell trajectories, ensuring smooth and accurate velocity reconstruction.
Comprehensive simulation studies spanning a broad range of flow velocities—from slow capillary speeds around 1 mm/s up to 60 mm/s—demonstrate R-Flow’s robustness and accuracy. Critically, these simulations validate its angle independence, a notable advantage over conventional techniques whose accuracy deteriorates significantly with flow orientation. Phantom experiments further reinforce these findings, exhibiting strong correlations (correlation coefficients exceeding 0.8) with established reference measurements, underscoring R-Flow’s reliability.
Beyond theoretical validation, in vivo experiments underline the clinical relevance of R-Flow. Utilizing a chick embryo model, researchers successfully mapped lateral blood flow components, which are often undetectable by standard Doppler approaches due to angle limitations. This capability to detect multidirectional flow dynamics paves the way for nuanced vascular assessments in developmental biology and beyond.
Perhaps most compelling is R-Flow’s application in human liver imaging, where it generated high-sensitivity flow vector maps exhibiting correlations greater than 0.9 with reference standards. This level of precision is critical for evaluating microvascular health in hepatology, particularly in conditions like cirrhosis characterized by complex vascular remodeling and impaired perfusion.
In a rat model of liver cirrhosis, R-Flow revealed striking pathological changes in microcirculation, capturing perfusion transitions from healthy, evenly distributed multipath flows to diseased states dominated by directionally biased hemodynamics. These altered flow patterns correlated closely with the severity of fibrosis and hypoxia markers, suggesting that R-Flow could serve as a powerful noninvasive biomarker to monitor disease progression and therapeutic response.
The contrast-free nature of R-Flow not only enhances patient safety by obviating the need for injected agents but also facilitates repeated real-time imaging sessions, ideal for both clinical diagnostics and longitudinal research studies. The algorithmic framework lends itself well to integration with existing ultrasound platforms, promising broad accessibility and rapid clinical translation.
By coupling advanced mathematical transforms with state-of-the-art ultrafast ultrasound imaging, R-Flow represents a monumental stride toward precision medicine. It equips clinicians and researchers with unprecedented visibility into microvascular flow dynamics, a realm historically challenging to interrogate. This capability is poised to transform the management of vascular diseases, enabling earlier diagnosis, more informed therapeutic decisions, and better patient outcomes.
Looking ahead, the research team envisions expanding R-Flow’s applications across various organ systems and disease states, including oncology where tumor microvasculature patterns critically influence prognosis and treatment. Further, with ongoing enhancements in computational efficiency, R-Flow may soon support bedside, real-time flow mapping, bringing high-resolution vascular insights directly into clinical workflows.
The advent of R-Flow underscores the power of interdisciplinary innovation, where engineering principles, physics, and clinical science converge to tackle unmet medical needs. As this technology gains momentum, it stands to redefine vascular imaging standards and unlock new frontiers in understanding blood flow’s role in health and disease.
Subject of Research: Not applicable
Article Title: Mapping Microvascular Flow via Radon Transform Ultrasound: Technical Advances and Pilot Application
News Publication Date: 24-Feb-2026
Web References: http://dx.doi.org/10.34133/bmef.0234
Image Credits: Chengwu Lab@MCCMS & Shigao Lab@MCCMS
Keywords: Ultrasound, Imaging, Blood flow, Lung cancer