In a groundbreaking study published in Translational Psychiatry, researchers Y. Huo, W. Huang, Z. Liu, and colleagues unveil new insights into the intricate patterns of brain aging linked to Alzheimer’s disease. Their work, entitled “Functional system-specific brain aging across the Alzheimer’s disease continuum,” leverages state-of-the-art neuroimaging and analytical techniques to dissect how distinct brain networks deteriorate over the course of the disease. As Alzheimer’s continues to impose a mounting global health burden, this study represents a significant step forward in understanding the nuanced progression of functional decline, potentially paving the way for targeted interventions.
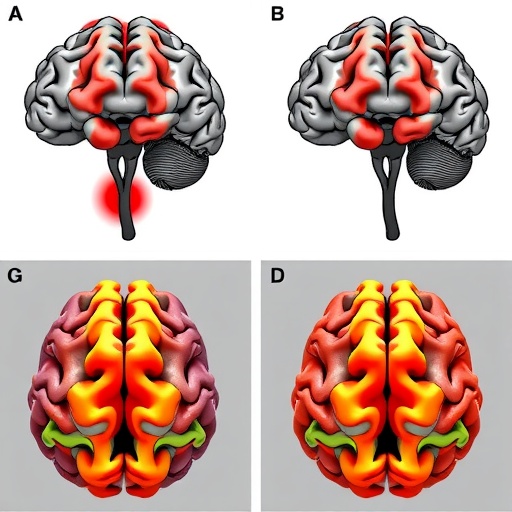
The research centers on the concept that brain aging in Alzheimer’s is not a uniform process but instead unfolds distinctly across various functional systems. This systemic perspective departs from traditional views that often consider the brain as a whole when examining neurodegeneration. By identifying patterns specific to networks such as the default mode, executive control, and salience networks, the authors provide compelling evidence that Alzheimer’s-related functional decline follows pathways aligned with the brain’s intrinsic organizational architecture.
Employing a longitudinal cohort study design complemented by cross-sectional analyses, the investigators analyzed neuroimaging data from subjects spanning cognitively healthy individuals to those in advanced stages of Alzheimer’s. Advanced functional MRI (fMRI) methodologies enabled the team to quantify changes in connectivity strength and network efficiency. Their approach intricately maps how the neurodegenerative cascade selectively impacts brain circuits, offering unprecedented resolution in characterizing disease progression over time.
One of the study’s pivotal findings is the differential vulnerability of brain networks. The default mode network, long implicated in memory and self-referential cognition, exhibited early and substantial functional degradation. In contrast, networks associated with sensory processing remained comparatively preserved until later disease stages. This dynamic suggests that Alzheimer’s pathology initially targets high-order cognitive systems before spreading to more basic perceptual functions, resonating with clinical observations of cognitive symptomatology.
This network-specific aging pattern was further corroborated by measures of brain atrophy and amyloid-beta accumulation derived from complementary neuroimaging modalities. By integrating multimodal imaging data, the researchers were able to correlate functional impairments with structural damage and pathological protein deposition, offering a holistic view of Alzheimer’s pathophysiology that transcends isolated biomarker analysis.
Intriguingly, the study also demonstrates that the rate of functional decline within each network correlates with distinct clinical phenotypes and cognitive trajectories. For instance, accelerated deterioration in the executive control network was linked to pronounced deficits in attention and problem-solving skills, while alterations in the salience network aligned with emotional dysregulation. This fine-grained mapping of symptoms to network-level dysfunction underscores the potential for precision medicine approaches tailored to individual functional profiles.
The methodological rigor extends into the use of novel computational models designed to quantify “brain age” at a network-specific scale. Unlike traditional brain aging models that provide a global estimate, these refined algorithms yield multiple aging metrics corresponding to each functional system. Such granularity not only enhances sensitivity for early detection but also enables monitoring of therapeutic efficacy when interventions are applied, marking a leap forward for clinical neurology.
From a translational perspective, these insights bolster the rationale for developing targeted neuroprotective strategies. If certain networks are selectively vulnerable during distinct disease phases, pharmaceutical and neuromodulatory interventions can be optimized to protect or restore functions specific to those systems. This could revolutionize treatment paradigms for Alzheimer’s, which historically have struggled against the complexity and heterogeneity of the disease.
Moreover, this study challenges the field to reconsider diagnostic frameworks by incorporating network aging profiles alongside conventional biomarkers. The researchers propose that future Alzheimer’s diagnostic criteria might integrate system-specific brain age metrics as adjunctive markers, enhancing diagnostic accuracy and prognostic precision. This shift would facilitate earlier and more personalized therapeutic decisions, ultimately improving patient outcomes.
Equally important is the implication for cognitive rehabilitation. Understanding which networks are impaired and at what stages opens avenues for designing rehabilitation protocols focusing on network-specific cognitive domains. Clinical interventions could become more adaptive and responsive, tailored to the evolving neurofunctional landscape of individuals living with Alzheimer’s.
The interdisciplinary collaboration underpinning this research spans neurology, neuroimaging, computational neuroscience, and psychiatry, reflecting the complex nature of Alzheimer’s disease research today. It exemplifies how integrating expertise from diverse fields can accelerate discovery and model the brain’s complexity with greater fidelity, a critical endeavor in combating neurodegeneration.
Looking ahead, the authors emphasize the necessity for further studies incorporating larger and more diverse cohorts to validate and refine their network-specific aging models. They advocate for longitudinal monitoring to chart disease trajectories across populations, as well as experimental interventions to test the reversibility or mitigation of network dysfunction. This trajectory promises to deepen our understanding of brain aging mechanisms and transform clinical practice.
This research heralds a new era where brain aging is understood through the lens of functional systems rather than global metrics, offering nuanced perspectives on Alzheimer’s disease progression. By highlighting specific vulnerability patterns and their clinical correlates, the work sets the stage for breakthroughs in diagnostics, treatment, and rehabilitation that could fundamentally alter the landscape of neurodegenerative disease management.
In sum, the study by Huo et al. constitutes a monumental stride in unraveling the complex tapestry of Alzheimer’s disease. Through innovative neuroimaging and analytical approaches, it elevates our comprehension of brain aging from a monolithic process to a multifaceted phenomenon, intricately tied to the brain’s functional architecture. The implications for personalized medicine, early intervention, and improved patient care are profound, signaling hope in the fight against this formidable disease.
As the scientific community continues to grapple with Alzheimer’s complexities, research like this underscores the critical importance of detailed functional analyses. They not only illuminate the biology underpinning cognitive decline but also point the way toward tangible therapeutic advances that could improve millions of lives worldwide. This breakthrough exemplifies the power of cutting-edge neuroscience to rewrite our understanding and, ultimately, our response to neurodegenerative disease.
Subject of Research: Functional system-specific brain aging mechanisms across the Alzheimer’s disease continuum
Article Title: Functional system-specific brain aging across the Alzheimer’s disease continuum
Article References:
Huo, Y., Huang, W., Liu, Z. et al. Functional system-specific brain aging across the Alzheimer’s disease continuum. Transl Psychiatry (2026). https://doi.org/10.1038/s41398-026-04081-8
Image Credits: AI Generated