Plant immunity represents a fascinating paradigm of autonomous cellular defense mechanisms unlike the more centralized immune systems typically seen in animals. Each plant cell independently detects invading pathogens and orchestrates an immune response, simultaneously engaging in systemic communication across the entire organism. This decentralized system reveals elegant molecular strategies plants have evolved, centered around the detection of intracellular threats through sophisticated receptor proteins known as nucleotide-binding leucine-rich repeat (NLR) immune receptors.
NLR receptors play a pivotal role in recognizing pathogen-derived effector proteins, which are injected directly into plant cells by pathogens to suppress immune responses and manipulate host cellular processes. Unlike plasma membrane-bound surface receptors, NLRs operate inside the cell, continuously surveilling the internal milieu for biochemical signatures indicative of pathogenic activity. These large, modular proteins feature three core domains: a central nucleotide-binding domain essential for activation, a leucine-rich repeat (LRR) domain responsible for ligand recognition or autoregulation, and a variable N-terminal domain, which classifies NLRs into two principal groups based on its structure—TIR (Toll/interleukin-1 receptor-like) or CC (coiled-coil) types.
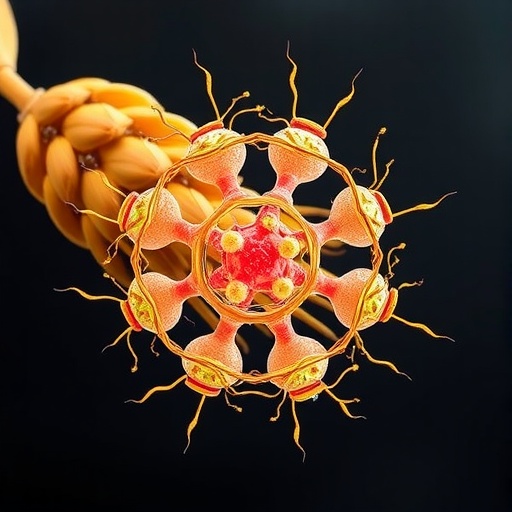
Upon activation, NLR proteins undergo dramatic conformational changes, often oligomerizing into higher-order assemblies termed resistosomes. These multi-subunit complexes serve as molecular machines that transduce pathogen detection signals into immune responses. Previously characterized resistosomes, such as ZAR1 and Sr35, typically form pentameric structures, whereas others like NRC2 and NRC4 assemble into hexamers, both functioning as calcium-permeable channels that facilitate Ca^2+ influx into the cytoplasm—a critical early signaling event in plant immunity.
Intriguingly, the CC_G10 subclass of CC-NLRs remained enigmatic with respect to their activation dynamics and oligomeric structures despite their clear functional importance. A groundbreaking study led by Prof. LIU Zhiyong and colleagues at the Institute of Genetics and Developmental Biology of the Chinese Academy of Sciences, in collaboration with researchers from The Sainsbury Laboratory and other renowned institutions, has unveiled the first octameric resistosome formed by a wheat CC_G10-NLR immune receptor called WAI3. This discovery not only expands the structural repertoire of resistosomes but also underscores previously unappreciated complexity in plant immune signaling architectures.
The initial clue for this breakthrough came from the analysis of a spontaneous autoimmune mutant in wheat, named M3045, derived from the “Zhongke 331” cultivar. The mutant exhibited autoactivation of immune responses leading to deleterious effects on growth, a trade-off reflecting hyperactive defense at the expense of development. Through meticulous map-based cloning, the team identified the causal gene, Wheat Autoimmunity 3 (WAI3), encoding a CC_G10-NLR protein harboring a gain-of-function mutation within its LRR domain. This mutation effectively bypasses the regulations that normally keep NLR activation tightly controlled.
Expressing the WAI3 protein in Nicotiana benthamiana, a model plant system extensively used for heterologous expression, allowed the researchers to purify sufficient protein and resolve its activated resistosome structure using cryo-electron microscopy (cryo-EM). Remarkably, WAI3 assembles into an octameric ring-shaped complex, distinct both in stoichiometry and conformation from previously described pentameric or hexameric resistosomes. This unique oligomeric arrangement reveals an unanticipated mode of NLR activation and immune signal propagation within plant cells.
Functional assays further demonstrated that the WAI3 resistosome acts as a calcium channel, mediating Ca^2+ influx into the cytoplasm—a hallmark signaling event triggering downstream immune pathways. Importantly, when expressed in animal cells, the WAI3 resistosome failed to generate comparable calcium currents, implying that yet unknown plant-specific cofactors or lipid membrane compositions might be required for full functional activity. This observation parallels previous findings with other CC-NLRs like NRC4 that also necessitate plant cellular contexts for optimal function.
Beyond wheat, the researchers extended their quest to the model dicot Arabidopsis thaliana, focusing on the homologous CC_G10-NLR protein RPS2. Although difficulties in obtaining pure protein for cryo-EM precluded direct structural elucidation, functional experiments confirmed that activated RPS2 similarly oligomerizes into an octameric resistosome and mediates calcium influx. This cross-species conservation underscores an evolutionary conserved mechanism in plant immunity spanning both monocots and dicots, illustrating a fundamental biological principle in the plant kingdom.
Taken together, this study revolutionizes our understanding of plant NLR immune receptors by revealing an entirely new class of resistosomes with octameric architecture, expanding the functional and structural diversity of these immune complexes. The findings emphasize the invaluable role of wheat as a genetic and biochemical model for plant immunity research, despite the traditional focus on Arabidopsis and other model species. This work not only propels fundamental plant biology forward but may also inspire novel strategies for engineering durable disease resistance in crops through targeted manipulation of CC_G10-NLR pathways.
The research paper, published in the prestigious journal Cell, meticulously dissects the molecular details of WAI3 activation, resistosome assembly, and ion channel function using state-of-the-art structural biology techniques combined with functional validation across plant species. By illuminating the unique assembly mechanism of the CC_G10 resistosome, the study opens new avenues for dissecting the intricate signaling networks that sustain plant immunity, potentially informing next-generation agricultural technologies aimed at bolstering crop resilience in an era of mounting biotic stresses.
This landmark discovery represents a significant leap forward in the plant immunity field, providing the first direct structural evidence of an octameric NLR resistosome and the functional dynamics of its calcium channel activity. The collaborative effort exemplifies how integrative approaches spanning genetics, structural biology, and physiology can unravel previously cryptic biological phenomena, further bridging molecular plant science with translational applications in sustainable agriculture.
Subject of Research: Plant Immunity, NLR Immune Receptors, Resistosome Structure, Calcium Signaling
Article Title: An activated wheat CCG10-NLR immune receptor forms an octameric resistosome
News Publication Date: 20-Mar-2026
Web References:
https://doi.org/10.1016/j.cell.2026.02.024
Keywords: Plant immunity, NLR receptors, CC_G10-NLR, resistosome, octameric complex, calcium influx, wheat, Arabidopsis thaliana, cryo-electron microscopy, immune signaling