Recent advancements in biomedical sciences have shed light on the intricate mechanisms governing cellular behavior in response to their physical environment. A study conducted by Huang and colleagues, published in the Journal of Biomedical Science, focuses on the regulation of a specific long non-coding RNA known as Neat1 and a protein called PSPC1 in renal progenitor cells subjected to varying substrate stiffness. This research opens new avenues for manipulating cellular fate, which could have significant implications for regenerative medicine and tissue engineering.
The relationship between substrate stiffness and cell fate is a profound area of inquiry that intertwines biophysics with cellular biology. Substrate stiffness refers to the rigidity of the material that cells grow on, which can profoundly influence various cell functions, including growth, differentiation, and gene expression. The TGF-β1 pathway, a crucial signaling pathway in many cellular processes, including fibrosis and repair, was highlighted in this research. The team investigated how changes in substrate stiffness alter the response of renal progenitor cells when exposed to TGF-β1.
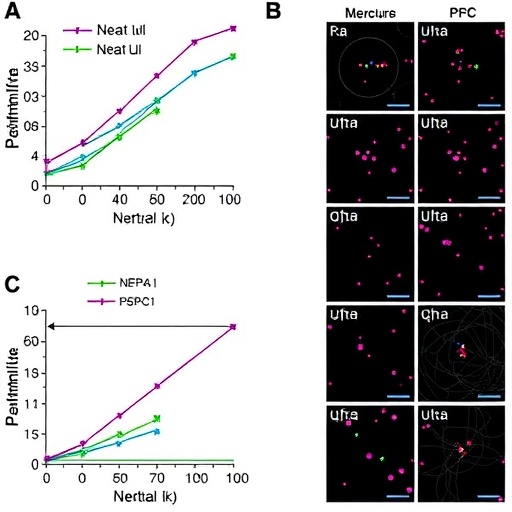
The authors of the study first established the importance of Neat1 in the cellular nucleus. Neat1 is known to be involved in the formation of nuclear paraspeckles, which are specific subnuclear structures associated with the regulation of gene expression and cellular stress responses. The research presented evidence that substrate stiffness could modulate the expression levels of Neat1, thereby influencing the cells’ transcriptional landscape and their subsequent behaviors.
A unique aspect of this study is the focus on PSPC1, a protein that plays a pivotal role in the organization of paraspeckles in the nucleus. The researchers demonstrated that varying substrate stiffness not only affected Neat1 expression but also altered PSPC1 localization and function within the cell. This connection provides a critical link between the biomechanical properties of the extracellular matrix and the molecular dynamics within the nucleus.
Understanding how mechanical cues translate into biological responses at the molecular level is essential for developing effective strategies for tissue regeneration. The findings from Huang and colleagues suggest that by manipulating substrate stiffness, it may be possible to control the differentiation pathways of renal progenitor cells. Such control is vital for optimizing cell therapies aimed at kidney repair and regeneration, especially in the context of renal diseases where cellular dysfunction plays a central role.
Additionally, the implications of these findings extend beyond renal progenitor cells. The general principles of mechanotransduction—how cells sense and respond to mechanical stimuli—could be applied to other cell types and tissues. This could pave the way for developing novel biomaterials that can better mimic the physiological conditions of the tissue they aim to replace or repair.
The complexity of the TGF-β1 signaling pathway adds another layer of intrigue to the study. TGF-β1 is known for its dual role in promoting fibrosis and contributing to tissue regeneration. By elucidating how Neat1 and PSPC1 interact within this pathway, the research offers valuable insights into how renal progenitor cells can be directed towards desired outcomes, whether it be healing or fibrosis.
As research in this domain continues to progress, there are several factors to consider. For instance, the interplay of other mechanical properties—such as topography and density—along with stiffness may yield further insights into cell behavior. Future studies could involve exploring these parameters in conjunction with Neat1 and PSPC1 to create a more comprehensive understanding of the mechanical microenvironment’s influence on cellular activities.
The potential clinical applications of such research are profound. For instance, insights gained from this study could inform the design of advanced scaffolds for kidney tissue engineering that not only support cell attachment and growth but also instruct cells towards a specific fate through controlled mechanical properties. This approach could dramatically enhance the efficacy of tissue engineering strategies aimed at restoring kidney function following injury or disease.
In summary, the research conducted by Huang et al. highlights the critical role of substrate stiffness in regulating the expression of Neat1 and PSPC1 in renal progenitor cells under the influence of TGF-β1. As the scientific community delves deeper into the mechanisms of mechanotransduction, these findings may catalyze a new wave of therapeutic strategies aimed at harnessing cellular responses for regenerative medicine. The synergy between mechanical cues and molecular biology is a promising frontier that could redefine our approaches to treating complex diseases.
The study lays a foundation for future research exploring the various dimensions of cellular response to mechanical stimuli. By continuing to unravel the complexities of how physical forces shape cellular destiny, scientists are paving the way for innovative solutions that could revolutionize medical treatments for a range of conditions. The implications of this research are boundless, echoing the essential role of biomechanics in cellular function and highlighting the need for interdisciplinary collaboration to explore these critical connections further.
In conclusion, the careful investigation led by Huang and collaborators not only adds a vital piece to the puzzle of cell behavior in response to mechanical environments but also sparks curiosity for further studies that could enhance our understanding of tissue engineering and regenerative medicine. The journey from basic science to clinical application remains long, yet studies like this are crucial in bridging that gap, ensuring that theoretical discoveries transform into tangible health benefits for future generations.
Subject of Research: Mechanobiology of renal progenitor cells and their regulation by substrate stiffness.
Article Title: Regulation of the mechanoresponsive Neat1 and PSPC1 by substrate stiffness in TGF-β1-induced renal progenitor cell fate.
Article References: Huang, HN., Lee, LW., Kuo, CH. et al. Regulation of the mechanoresponsive Neat1 and PSPC1 by substrate stiffness in TGF-β1-induced renal progenitor cell fate. J Biomed Sci 32, 99 (2025). https://doi.org/10.1186/s12929-025-01196-w
Image Credits: AI Generated
DOI: https://doi.org/10.1186/s12929-025-01196-w
Keywords: Neat1, PSPC1, substrate stiffness, renal progenitor cells, TGF-β1, mechanotransduction, tissue engineering, regenerative medicine.