In recent years, the quest for efficient and sustainable hydrogen production has gained prominence, primarily driven by the need for renewable energy sources. A critical aspect of this process lies in the electrolysis of water, which facilitates the conversion of electrical energy into chemical energy in the form of hydrogen. This hydrogen, when generated from renewable energy, is carbon-neutral and regarded as a pivotal element in transitioning towards sustainable energy systems. Unfortunately, the process of water electrolysis faces significant challenges, particularly concerning the oxygen evolution reaction (OER) at the anode. This reaction tends to slow down the overall rate of hydrogen production, underscoring the need for efficient catalysts to enhance this reaction.
To address this challenge, scientists have been exploring various materials to improve the efficiency of catalysts used in the OER process. Among them, nickel-based compounds have emerged as promising candidates due to their cost-effectiveness and favorable catalytic properties. In a groundbreaking study led by Dr. Prashanth Menezes and his research team, the potential of a unique class of materials known as clathrates is being explored as catalysts for the OER. These materials, specifically made from a combination of barium, nickel, and germanium, offer a fascinating crystalline structure characterized by polyhedral cages. Their intricate composition provides special properties that could revolutionize the electrolysis process.
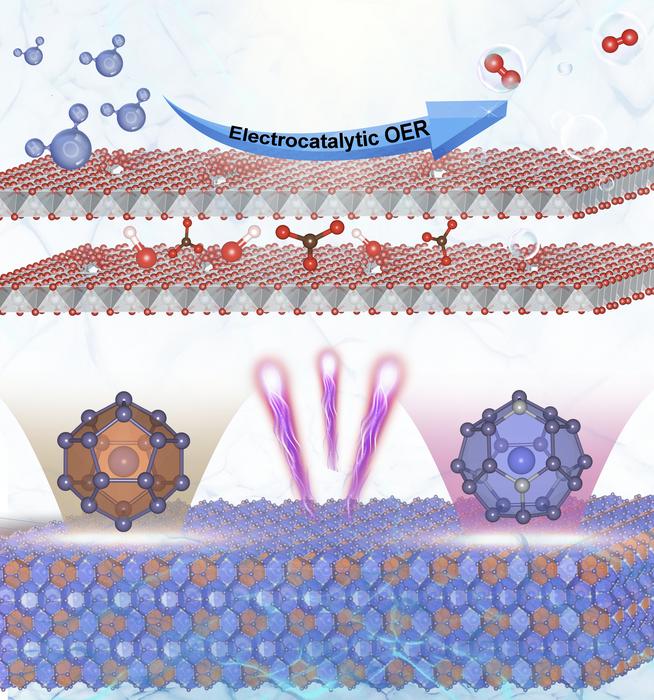
The research focuses on a specific clathrate compound, Ba₈Ni₆Ge₄₀, produced at the Technical University of Munich. The unique structure of clathrates, which consists of interlocked cages formed by nickel and germanium that enclose barium, presents an intriguing opportunity for catalysis. Traditionally, the surface area of nickel-based catalysts is limited, which restricts their efficiency in facilitating the OER. Dr. Menezes and his team hypothesized that leveraging the structural properties of clathrates could yield a more effective catalyst.
In a series of electrochemical experiments, the Ba₈Ni₆Ge₄₀ catalyst exhibited remarkable performance, surpassing the efficiency of conventional nickel-based catalysts at a current density of 550 mA cm⁻². This specific current density is significant, as it aligns with conditions typically encountered in industrial electrolysis applications. Notably, the stability of this catalyst was commendable; after ten days of continuous operation, the activity levels remained stable, highlighting the potential for practical applications in sustainable hydrogen production.
To unravel the mechanisms behind this enhanced performance, the research team utilized a combination of advanced experimental techniques. In situ X-ray absorption spectroscopy (XAS) studies conducted at BESSY II, a synchrotron facility, provided valuable insights into the behavior of the clathrate materials under operational conditions. The analysis illuminated a crucial transformation occurring within the Ba₈Ni₆Ge₄₀ particles when they were placed in an aqueous electrolyte and subjected to an electric field.
The findings revealed that the germanium and barium atoms, which constitute a significant portion of the clathrate structure, dissolved from the framework under the applied electric field. This structural transformation left behind a highly porous, sponge-like network comprised almost entirely of nickel, notably increasing its surface area. As Dr. Niklas Hausmann from Menezes’ team explained, this transformation facilitates a greater interaction between the catalytically active nickel centers and the electrolyte, thereby enhancing the efficiency of the OER process.
The researchers were pleasantly surprised by the exceptional performance exhibited by these clathrate-derived catalysts. They foresee potential applications extending beyond the Ba₈Ni₆Ge₄₀ compound, anticipating that similar results could emerge from other transition metal clathrates that may also serve as effective electrocatalysts. The implications of this discovery are profound, as it opens up new avenues in the search for materials that can efficiently catalyze water splitting reactions, potentially reshaping the landscape of renewable energy production.
In summary, the innovative approach of utilizing clathrates as catalysts could lead to significant advancements in hydrogen production via water electrolysis. The structural advantages offered by these materials, coupled with their resilience and efficiency, make them exceptionally appealing for industrial applications. As the demand for sustainable energy solutions continues to escalate, the significance of such research becomes increasingly evident. The collaboration between fundamental research and practical applications holds the key to transforming the energy landscape and fostering a more sustainable future.
The developments highlighted in this study represent a considerable leap forward in materials science, catalysis, and renewable energy technologies. By pushing the boundaries of what is known and exploring unconventional materials, researchers like Dr. Menezes and his team are paving the way for innovations that have the potential to alter our approach to energy production and utilization. The ongoing investigation into clathrate-based catalysts promises not only to enhance the efficiency of oxygen evolution but also to contribute to the broader goal of achieving a sustainable and carbon-neutral energy future.
As the scientific community continues to delve deeper into the properties and applications of clathrates, the prospects of unlocking new, high-performing catalytic systems become increasingly viable. With such advancements, the future of hydrogen production looks promising, positioned to play a crucial role in the development of a more sustainable energy ecosystem for generations to come.
Subject of Research: Clathrate compounds as catalysts for the oxygen evolution reaction
Article Title: a-Ni-Ge Clathrate Transformation Maximizes Active Site Utilization of Nickel for Enhanced Oxygen Evolution Performance
News Publication Date: 26-Mar-2025
Web References: DOI
References: None provided
Image Credits: Hongyuan Yang/HZB/TUB
Keywords
Sustainable hydrogen production, electrolysis, oxygen evolution reaction, nickel-based catalysts, clathrates, Ba₈Ni₆Ge₄₀, electrochemical efficiency, renewable energy.