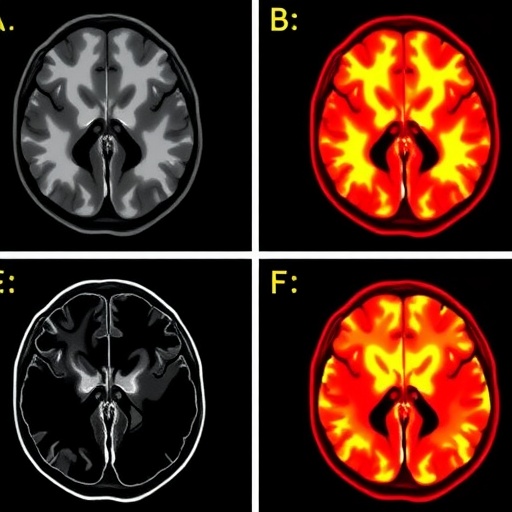
A groundbreaking study published in Molecular Psychiatry on March 5, 2026, has unveiled critical molecular insights into ketamine’s rapid antidepressant effects in patients with treatment-resistant depression (TRD). Despite ketamine’s emergence as a transformative therapeutic option for individuals unresponsive to conventional antidepressants, the precise mechanisms underlying its efficacy in the living human brain have long eluded researchers. By harnessing an innovative positron emission tomography (PET) tracer, [¹¹C]K-2, the research team led by Professor Takuya Takahashi at Yokohama City University has, for the first time, dynamically visualized alterations in cell-surface α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor (AMPAR) density—a pivotal glutamatergic synaptic receptor—in patients undergoing ketamine treatment.
Major depressive disorder is a debilitating global health crisis, with an estimated 30% of patients developing TRD that defies standard pharmacological intervention. Ketamine, an NMDA receptor antagonist, has revolutionized the antidepressant landscape by producing rapid symptom remission within hours to days, yet its molecular impact on neural circuits remained enigmatic. Previous preclinical models implicated AMPA receptors in mediating these antidepressant responses through synaptic potentiation and plasticity enhancement, but direct human evidence was lacking. The advent of the selective radioligand [¹¹C]K-2 allowed Takahashi’s team to overcome this barrier by enabling in vivo visualization of AMPARs in the human brain, illuminating ketamine’s receptor-level dynamics.
The clinical investigation incorporated 34 patients diagnosed with TRD and 49 demographically matched healthy controls, across three rigorously designed clinical trials in Japan. Participants underwent two weeks of either intravenous ketamine or placebo administration, with PET scans conducted prior to treatment onset and following the final infusion session. This longitudinal design permitted precise mapping of ketamine-induced receptor density changes alongside concurrent clinical symptomatology assessments, revealing complex neurochemical remodeling in responsive patients.
Contrary to a uniform receptor modulation hypothesis, results demonstrated highly region-specific patterns of AMPAR density alteration in patients with TRD. Baseline scans revealed extensive dysregulation of AMPAR throughout cortical and subcortical structures when compared to healthy brains, signifying foundational glutamatergic impairments in depression. Post-ketamine imaging disclosed that symptom improvement correlated with pronounced increases in AMPAR density within selective cortical regions known for higher-order cognitive and affective functions. Simultaneously, decreases in AMPAR density were identified in the habenula, a critical node in reward processing and aversive signaling, highlighting an intricate bidirectional regulation linked to therapeutic outcomes.
This spatially nuanced modulation underlines ketamine’s ability to recalibrate neural circuit dynamics by selectively enhancing excitatory synaptic signaling where deficits prevail, while dampening overactive pathways implicated in maladaptive reward responses. The findings support a model wherein ketamine’s antidepressant efficacy emanates from its capacity to rebalance glutamate receptor distribution and synaptic plasticity across discrete brain networks, thereby restoring functional connectivity and alleviating depressive symptoms rapidly.
Professor Takahashi emphasized, “Our application of [¹¹C]K-2 PET imaging marks a transformational leap in human neuropsychiatric research by directly linking molecular receptor dynamics to clinical antidepressant response in TRD. These insights bridge a critical translational gap from animal studies to human therapeutics.” By empirically verifying AMPAR’s central role in ketamine action, this work substantiates decades of rodent preclinical findings and elevates the receptor as a promising target for future drug development efforts aimed at precision psychiatry.
Beyond advancing fundamental neuroscience, the utility of AMPAR PET imaging as a biomarker harbors significant clinical implications. It offers a novel, objective method for monitoring treatment efficacy, potentially enabling clinicians to predict which patients will benefit from ketamine prior to initiation, thereby avoiding trial-and-error prescribing and reducing patient burden. This precision approach aligns with modern trends in personalized medicine, addressing urgent unmet needs in mental health care where heterogeneity in drug response hampers effective management.
The study’s robust methodology, combining longitudinal imaging with comprehensive clinical phenotyping, sets a new standard for integrative neuroimaging research in psychiatry. It also accentuates the importance of interdisciplinary collaboration spanning radiochemistry, neuropharmacology, psychiatry, and clinical imaging to unravel complex brain disorders at a molecular level. Such integration can expedite the translation of biological discoveries into actionable medical interventions.
Funding for this landmark research was provided by multiple Japanese scientific agencies and foundations, including the Ministry of Education, Culture, Sports, Science and Technology, the Japan Agency for Medical Research and Development (AMED), and the Japan Society for the Promotion of Science KAKENHI, among others. Importantly, ethical compliance and regulatory approvals were strictly adhered to, ensuring patient safety and data integrity throughout the clinical trials.
Looking ahead, Professor Takahashi and his team envision expanding the application of [¹¹C]K-2 PET imaging to other neuropsychiatric disorders characterized by glutamatergic dysregulation, as well as exploring novel pharmacological agents targeting AMPAR dynamics. The prospect of tailoring antidepressant therapies based on receptor-level biomarkers heralds a new paradigm in mental health treatment, promising improved outcomes and accelerated recovery for patients worldwide.
In summary, this pioneering investigation elucidates the dynamic and regionally specific modulation of AMPA receptors as a core mechanism underpinning ketamine’s rapid antidepressant efficacy in treatment-resistant depression. The marriage of cutting-edge molecular imaging with clinical psychiatry not only deepens our understanding of depression’s neurobiology but also catalyzes the development of personalized, mechanism-based therapies destined to transform psychiatric practice.
Subject of Research: People
Article Title: The dynamics of AMPA receptors underlies the efficacy of ketamine in treatment resistant patients with depression
News Publication Date: 5-Mar-2026
Web References: https://10.0.4.14/s41380-026-03510-w
References: DOI: 10.0.4.14/s41380-026-03510-w
Image Credits: Professor Takuya Takahashi from Yokohama City University Graduate School of Medicine, Japan, and Dr. Hiroyuki Uchida from Keio University School of Medicine, Japan
Keywords: Depression, Mental health, Positron emission tomography, Biomarkers, Neuroscience, Glutamates, Clinical trials, Antidepressants