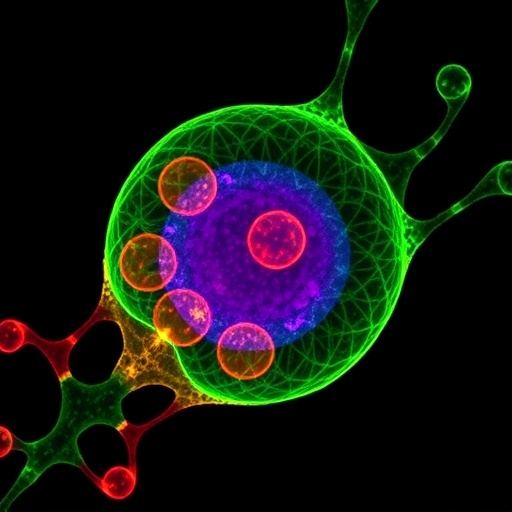
In a breakthrough poised to reshape cellular biology research, scientists have unveiled a pioneering imaging technique that allows for the direct visualization of individual lipid molecules within living mammalian cells. This advancement overcomes one of the most stubborn obstacles in the study of cellular membranes. Lipids, the fundamental building blocks of cell membranes, have long eluded detailed imaging due to their chemically elusive nature, leaving significant gaps in our understanding of their transport, localization, and dynamic behavior within cellular organelles.
Lipids perform a myriad of vital cellular functions beyond simply forming the structural matrix of membranes. They compartmentalize biochemical reactions, store energy, and play integral roles in intracellular signaling pathways. Given that every cell synthesizes thousands of distinct lipid species, ensuring the precise distribution and maintenance of these lipids across the diverse membrane systems of organelles is critical for cellular homeostasis. When lipid transport and balance go awry, metabolic syndromes and neurodegenerative diseases often follow, emphasizing the urgency of elucidating these mechanisms.
A collaborative effort spearheaded by researchers at the Max Planck Institute of Molecular Cell Biology and Genetics (MPI-CBG) in Dresden, Germany, together with experts from TUD Dresden University’s Biotechnology Center and the Helmholtz-Zentrum Dresden-Rossendorf (HZDR), has culminated in a novel chemical labeling approach coupled with advanced fluorescence microscopy. This method empowers scientists to track native-like lipid molecules inside living cells at unprecedented resolution. The interdisciplinary team synthesized bifunctional lipid analogs, structurally similar to their natural counterparts but modified to be photoactivatable under ultraviolet (UV) light, enabling spatial and temporal control over lipid-protein crosslinking.
The ingenuity lies in these chemically tailored lipids’ ability to integrate seamlessly into cellular membranes and, upon UV activation, to instantly attach covalently to proximate proteins. This “locking” of lipids in situ preserves their exact localization and interaction environment, which can be monitored using conventional fluorescence imaging techniques. Human bone and intestinal cell cultures were chosen as ideal model systems due to their compatibility with high-resolution microscopy and relevance to physiological contexts.
The spatial distribution of these artificial lipids revealed a complex and dynamic lipid trafficking network between the plasma membrane and intracellular organelles. The research team employed an innovative image analysis pipeline enhanced by artificial intelligence algorithms to segment cellular compartments accurately and quantify lipid transport kinetics. This computational approach allowed precise mapping of lipid flow, unveiling intricate pathways hitherto invisible to cell biologists.
Complementing microscopy, mass spectrometry analyses performed by collaborators at MPI-CBG provided molecular-level insights into lipid structural modifications during transport. Ultra-high-resolution mass spectrometry revealed subtle chemical alterations occurring as lipids traverse from one membrane environment to another, shedding light on the biochemical transformations underpinning lipid trafficking and membrane remodeling.
Remarkably, the study demonstrated that the vast majority of lipid movement—between 85% and 95%—occurs via non-vesicular transport mechanisms facilitated by specialized lipid-transfer proteins. This paradigm challenges the long-standing assumption that vesicular trafficking dominates lipid distribution. Additionally, lipid transport by these carrier proteins was found to be up to ten times faster than vesicle-mediated mechanisms, emphasizing the efficiency and specificity of protein-facilitated lipid shuttling.
These findings provide compelling evidence that lipid-transfer proteins play a pivotal role in maintaining the distinct lipid compositions of organelle membranes, which is paramount for their functionality and identity. The ability to visualize and quantify lipid dynamics in real time opens myriad avenues for dissecting the biochemical logic of membrane biogenesis and adaptation.
Beyond fundamental cell biology, this technological leap carries profound implications for biomedical research. Dysregulated lipid metabolism is implicated in numerous diseases, including nonalcoholic fatty liver disease, neurodegeneration, and metabolic disorders. The new imaging platform offers a robust tool to elucidate how lipid imbalances emerge and propagate pathology at the cellular level, ultimately guiding the development of targeted therapeutic interventions.
The origin of this breakthrough is a story of persistence and innovation. André Nadler and Alf Honigmann, principal investigators at MPI-CBG and BIOTEC respectively, began contemplating solutions to the lipid imaging problem shortly after joining the institute in 2014-2015. Over nearly five years, they refined lipid probes and imaging protocols, culminating in 2019 with fluorescence images that distinctly delineated lipid distributions in living cells. The advent of artificial intelligence in image processing was serendipitous, enabling the high-throughput, quantitative analyses essential for robust conclusions.
Future challenges remain for the field, including the identification and characterization of individual lipid-transfer proteins responsible for transporting specific lipid species. Moreover, understanding the energetic requirements and regulation mechanisms governing lipid transfer is crucial to fully decoding membrane biology.
This transformative study, published in Nature, charts an exciting course for cell biology, merging synthetic chemistry, biophysics, computational imaging, and molecular biology to illuminate one of the cell’s most enigmatic components. As lipid imaging matures, it heralds a new frontier in both basic science and translational medicine, promising to unravel the complex lipid-mediated pathways underpinning health and disease.
Subject of Research: Cells
Article Title: Quantitative imaging of lipid transport in mammalian cells
News Publication Date: 20-Aug-2025
Web References:
http://dx.doi.org/10.1038/s41586-025-09432-x
Image Credits: Kristin Böhlig and Juan Iglesias-Artola / Nature (2025) / MPI-CBG
Keywords: lipid transport, fluorescence microscopy, non-vesicular transport, lipid-transfer proteins, membrane biology, lipid imaging, cell organelles, synthetic lipids, mass spectrometry, artificial intelligence, mammalian cells, lipid metabolism