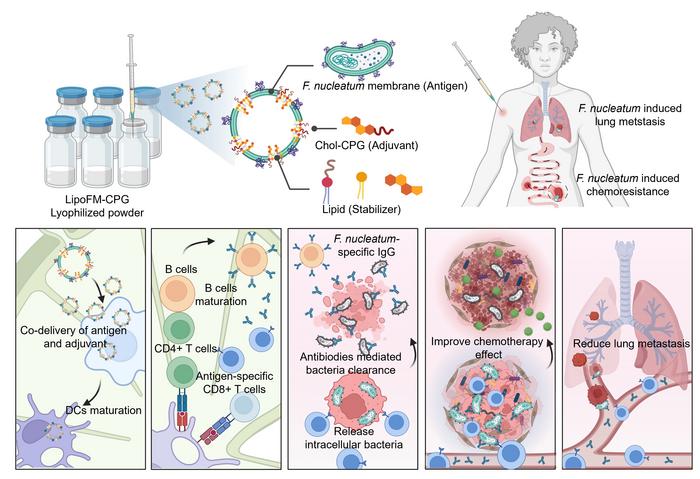
The human body hosts more than 1014 bacteria, which play crucial roles in human health. F. nucleatum, an oral anaerobe, is frequently associated with colorectal cancer and is linked to increased cancer cell proliferation, metastasis, and poor treatment outcomes. Recent studies have shown that F. nucleatum can trigger autophagy in cancer cells, leading to increased resistance to various chemotherapy drugs. Additionally, F. nucleatum promotes an immunosuppressive tumor microenvironment, hindering the infiltration of tumor-killing natural killer cells and T cells. Therefore, eliminating F. nucleatum within tumors could be a strategic approach to enhance therapeutic outcomes for colorectal cancer. Antibiotics are widely used to combat bacterial infections. However, they often indiscriminately kill both beneficial and harmful bacteria, potentially disrupting the intestinal microbiome. Bacterial vaccines offer a promising solution by targeting specific pathogens without harming the broader microbiome. However, developing effective bacterial vaccines is challenging due to the weak immunogenicity of major bacterial antigens. Although adjuvants can enhance the humoral response, effective vaccines require robust activation of antigen specific cellular response to eliminate intracellular pathogens. Hence, there is a strong desire for an antibacterial vaccine capable of eliciting robust immune responses to selectively eradicate F. nucleatum while preserving other microbiota.

Credit: ©Science China Press
The human body hosts more than 1014 bacteria, which play crucial roles in human health. F. nucleatum, an oral anaerobe, is frequently associated with colorectal cancer and is linked to increased cancer cell proliferation, metastasis, and poor treatment outcomes. Recent studies have shown that F. nucleatum can trigger autophagy in cancer cells, leading to increased resistance to various chemotherapy drugs. Additionally, F. nucleatum promotes an immunosuppressive tumor microenvironment, hindering the infiltration of tumor-killing natural killer cells and T cells. Therefore, eliminating F. nucleatum within tumors could be a strategic approach to enhance therapeutic outcomes for colorectal cancer. Antibiotics are widely used to combat bacterial infections. However, they often indiscriminately kill both beneficial and harmful bacteria, potentially disrupting the intestinal microbiome. Bacterial vaccines offer a promising solution by targeting specific pathogens without harming the broader microbiome. However, developing effective bacterial vaccines is challenging due to the weak immunogenicity of major bacterial antigens. Although adjuvants can enhance the humoral response, effective vaccines require robust activation of antigen specific cellular response to eliminate intracellular pathogens. Hence, there is a strong desire for an antibacterial vaccine capable of eliciting robust immune responses to selectively eradicate F. nucleatum while preserving other microbiota.
To address these challenges, in a recent research article published in Science Bulletin, a research team led by Professor Qian Chen from Soochow University (Institute of Functional Nano & Soft Materials, FUNSOM) proposed a novel bacterial vaccine (LipoFM-CPG) by inserting cholesterol-modified agonist CpG into F.nucleatum derived membranes. Compared with traditional emulsions with inactivated bacteria and Alum adjuvant, LipoFM-CPG co-delivers antigens and adjuvants, significantly enhancing dendritic cell maturation and antigen presentation. This stimulation leads to robust antibacterial cellular and humoral immune responses. The nanovaccine has demonstrated the ability to selectively and efficiently eradicate F. nucleatum, enhancing chemotherapy efficacy and reducing cancer metastasis in F. nucleatum-infected CRC. Importantly, compared to systemic or oral antibiotics, the nanovaccine has a negligible impact on the intratumoral and gut microbiota. Overall, the work highlights a strategy on designing a bacterial vaccine to elicit an immune response against intratumoral F. nucleatum, thereby potentiating cancer treatment, and this nanovaccine with simple manufacturing process, strong immunogenicity, and desired biocompatibility, should be a promising technology for selectively eliminating intratumoral bacteria and improving the therapeutic effect in bacteria-infected cancer.
###
See the article:
An emerging antibacterial nanovaccine for enhanced chemotherapy by selectively eliminating tumor-colonizing bacteria
Journal
Science Bulletin