For over a century and a half, ants have fascinated scientists and philosophers alike, serving as a living testament to the complexity of social behaviors and evolutionary innovation. From Aristotle’s admiration of their political organization to Daoist reflections on the transient nature of human affairs, ants have long symbolized order, diligence, and communal labor. This persistent intrigue transcended folklore, entering the realm of rigorous scientific inquiry with Darwin, who dedicated significant portions of his groundbreaking work, On the Origin of Species, to unraveling the paradox ants posed to natural selection theory. Specifically, Darwin was puzzled by the stark differences between ant workers—sterile and unable to reproduce—and their reproductive counterparts, the queens and males. How could such seemingly altruistic castes evolve when workers contributed no direct offspring?
Darwin’s theoretical resolution suggested that the workers indirectly propagated their genes by supporting their monogamous parents’ reproductive success. This hypothesis was revolutionary, introducing the concept that genes promoting altruistic behavior among siblings could be favored by natural selection. It implied that reproductive division of labor, with males contributing only sperm and queens fertilizing eggs across decades, enabled workers to specialize in brood care, colony defense, and foraging, sacrificing their own reproductive potential.
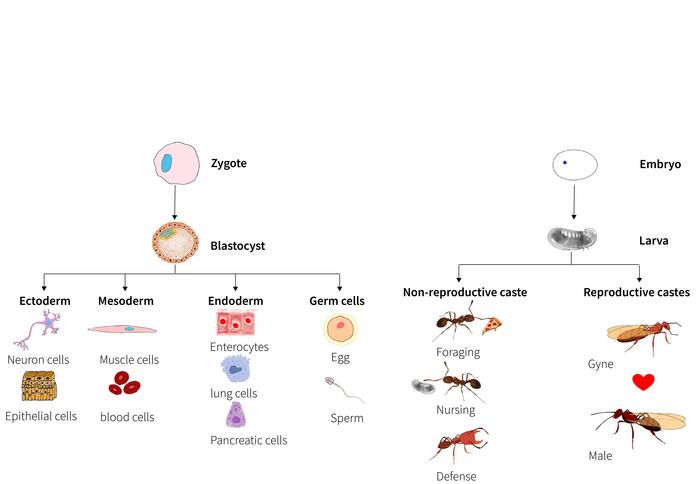
Since the 1960s, this conceptual framework has been substantiated through kin-selection theory. Ant colonies exemplify extended phenotypes, where the survival and reproductive output of related individuals—siblings—drive genetic success. The ergonomic efficiency of rearing siblings, optimized over millions of years, shaped the parallel developmental trajectories of queens and workers, encoded intimately within ant genomes. This fascinating parallel mirrors cellular differentiation in multicellular organisms, where a single genome orchestrates diverse cell types through regulated gene expression and developmental pathways.
Yet, the molecular and genomic underpinnings enabling such parallel caste differentiation in ants remained elusive. How could ant genomes be reorganized or functionally adapted to simultaneously produce divergent castes with radically distinct morphologies and behaviors? Addressing this enigma required a leap beyond fragmented genomic data, necessitating comprehensive sequencing efforts spanning diverse ant species worldwide. An ambitious project initiated almost a decade ago at the Section for Ecology and Evolution at the University of Copenhagen, now documented in a landmark publication in Cell, delivers unprecedented insights into the evolutionary rewiring of ant genomes.
This massive comparative genomics effort analyzed over 130 high-quality ant genomes, revealing that the genomic architecture of ants is largely dedicated to the origin and refinement of queen and worker castes. The data suggest an intricate evolutionary dynamic: after the initial emergence of ants during the late Jurassic period, natural selection sculpted their genomes in tandem with increases in colony complexity, including dramatic caste size dimorphisms and the appearance of specialized sterile soldiers. This genomic reorganization involved both large-scale reshuffling and conservation, illustrating the delicate balance between innovation and stability.
Remarkably, once ant genomes stabilized around the early Cretaceous, evolutionary pressures drove extensive gene reshuffling at a scale unparalleled in other animal lineages. This genomic plasticity appears closely linked to speciation rates across ant subfamilies, with subclades exhibiting higher gene rearrangement also showing greater species diversity. Meanwhile, smaller gene clusters implicated in regulating reproductive division of labor demonstrated remarkable conservation, persisting intact for over 100 million years. This dual phenomenon of genomic flexibility coupled with deep conservation provides a molecular canvas explaining both the immense diversification and functional specialization of ant superorganisms.
At the heart of this scientific breakthrough lies the pioneering collaboration between Professors Guojie Zhang and Jacobus (Koos) Boomsma. Both instrumental in early ant genome sequencing circa 2010-2011, they confronted the inherent limitations of fragmented data generated by the first generation of sequencing technologies. In response, in 2016, they launched the Global Ant Genomics Alliance (GAGA), harnessing advanced long-read sequencing technologies and bioinformatic innovations, fostering cooperation among global experts to amass extensive ant genomic resources. This consortium embodied a paradigm shift, enabling the assembly of highly contiguous genomes essential for unraveling complex genomic rearrangements governing caste differentiation.
Technological advances afforded by long-read sequencing overcame challenges posed by repetitive sequences and structural variants, which had previously hampered comprehensive analysis of ant genomes. The integration of these new datasets with ecological and evolutionary parameters facilitated robust correlations between genomic features and phenotypic traits such as colony size and caste morphology. This holistic approach exemplifies the intersection of cutting-edge genomics and classical evolutionary biology, unlocking secrets behind one of nature’s most successful social systems: ant supercolonies.
The study’s revelations extend beyond mere descriptive genomics, delving into evolutionary mechanisms that orchestrate social complexity. Genetic elements involved in task allocation, longevity differences between castes, and developmental pathways underlying morphological divergence were identified. The finding that genes regulating reproductive division of labor form stable clusters over immense evolutionary timescales highlights genomic modularity as a key factor sustaining social complexity. Conversely, the dynamic reshuffling of other genomic regions illustrates adaptive plasticity, allowing ants to fine-tune colony dynamics in response to ecological pressures.
Importantly, the conceptual analogy drawn between ant caste differentiation and cell specialization in multicellular organisms offers profound implications. It suggests that similar genomic strategies—such as parallel gene expression programs and epigenetic regulation—might underpin the emergence of biological complexity across scales, from cells within an organism to individuals within a superorganism. This perspective fosters cross-disciplinary dialogue, integrating evolutionary genomics with developmental biology and social ethology, and informs broader questions about the evolution of cooperation and division of labor.
Looking forward, the consortium’s dataset represents a treasure trove for future investigations. Functional analyses of caste-specific genes, epigenetic mechanisms modulating gene expression, and gene-environment interactions shaping social behavior constitute promising avenues. Furthermore, comparative studies with other eusocial insects such as bees and termites may elucidate convergent and divergent genomic strategies underpinning sociality. The insights gleaned may even inspire biotechnological applications, leveraging understanding of division of labor for innovations in robotics or synthetic biology.
This achievement exemplifies how long-term international collaboration, technological advancements, and integrative biology drive transformative discoveries. By deciphering the genomic blueprint of ant superorganisms, this research not only resolves a century-old evolutionary puzzle ignited by Darwin but also expands our comprehension of the molecular foundations of complex social systems. Ants, often overlooked despite their ecological ubiquity, now stand as a beacon illuminating fundamental principles of evolution, development, and cooperation.
For researchers, evolutionary biologists, and social scientists alike, these findings underscore the power of genomics to unlock nature’s intricate designs. They invite us to reconsider social insects not merely as colonies of individuals but as dynamic superorganisms whose genomes have been meticulously sculpted by natural selection to enable phenomenal integration and specialization. The ant genome thus serves as a model for understanding how genetic information can be organized to support extraordinary levels of biological and social complexity, offering profound insights with ramifications across the life sciences.
Subject of Research: Genomic organization underpinning caste differentiation and social complexity in ants.
Article Title: The Genomic Organization of Ant Superorganisms
Web References: https://db.cngb.org/antbase/project
Image Credits: Yuanhui Cai and Guo Ding
Keywords: Ant genomics, caste differentiation, social evolution, kin selection, genome rearrangement, division of labor, eusociality, evolutionary biology, long-read sequencing, superorganisms