In a groundbreaking study that bridges the fields of cancer biology, nutrition, and radiation medicine, researchers have unveiled intriguing insights into how calorie restriction (CR) modulates molecular pathways linked to cancer suppression in radiation-exposed mice. The latest research, soon to be published in BMC Cancer, sheds new light on the nuanced cellular mechanisms through which CR exerts its protective effects, particularly focusing on autophagy-related proteins and their interplay in the liver following radiation exposure.
Calorie restriction has long been recognized for its broad anti-cancer potential, demonstrating an ability to inhibit spontaneous tumor formation as well as chemically and radiation-induced carcinogenesis. Yet, the precise molecular and cellular underpinnings driving its efficacy have remained elusive. This new study dives deep into the biology of autophagy—a cellular housekeeping mechanism that maintains homeostasis by degrading and recycling cellular components—to understand how CR influences this critical process in the context of radiation-induced liver carcinogenesis.
Utilizing a well-established mouse model, the researchers exposed male B6C3F1 mice to either a sublethal dose of 3.8 Gy X-rays or no radiation at all during their early postnatal development. After this exposure at one week of age, the mice were assigned to two dietary regimens from seven weeks onward: a standard caloric intake or a calorie-restricted intake approximately 30% lower. This experimental design allowed the team to dissect the effects of both radiation and CR on liver pathology over an extended timeframe.
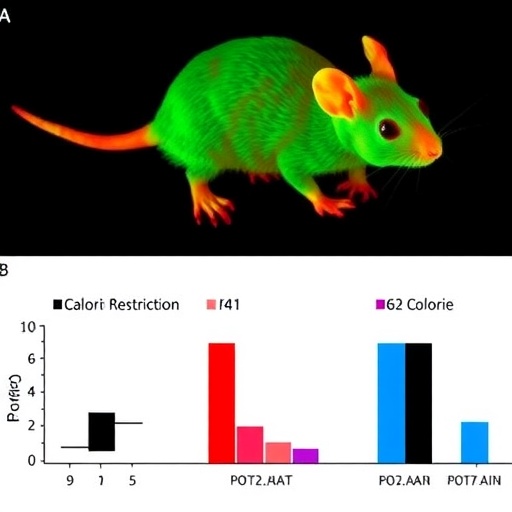
One of the pivotal findings of the study was that calorie restriction significantly curtailed the incidence of liver tumors in irradiated mice, reinforcing the notion of CR’s robust anti-carcinogenic capabilities under stress conditions like radiation exposure. While tumor suppression was observed, the researchers sought to elucidate whether autophagic activity might underpin this protective phenotype, focusing specifically on the proteins LC3 and p62, both key players in autophagosome formation and turnover.
Contrary to what might have been expected, LC3 levels in the liver remained relatively unchanged regardless of radiation or dietary intervention. LC3 is widely considered a canonical marker of autophagy induction, hence these data suggest that the overall autophagic flux was not significantly altered by CR in this context. This finding challenges simplistic views that increased autophagy uniformly mediates the cancer-suppressive effects of calorie restriction.
More striking, however, was the unprecedented behavior of the protein p62 (also known as SQSTM1), which serves as a selective autophagy receptor and signaling hub integrating stress responses. The investigators reported a significant and transient increase in p62 expression exclusively in the irradiated mice subjected to calorie restriction—the very group demonstrating suppressed tumorigenesis. This selective elevation in p62 expression hints at a sophisticated regulatory mechanism beyond canonical autophagy modulation.
Through detailed immunofluorescence analyses, the team observed that p62 was strongly expressed throughout the cytoplasm of hepatic cells in calorie-restricted mice. Intriguingly, some of these p62-overexpressing cells exhibited hallmarks of cell death, such as nuclear disappearance and reduced DNA content, which did not correlate with markers of cell proliferation. This suggests that p62 may be intimately linked to the elimination of damaged or potentially precancerous cells via a previously uncharacterized pathway, acting as a critical mediator in maintaining liver integrity after radiation insult.
The discovery of distinct populations of p62-high cells with diminished or absent nuclei exclusively under calorie restriction points to a novel, non-autophagic role of p62 in facilitating cellular turnover and carcinogenesis suppression. These insights raise provocative questions about whether p62 acts as a sensor or executor of damaged cell clearance in the liver’s microenvironment following radiation exposure.
Long-term CR, as monitored in this study, did not prompt substantial changes in overall autophagic activity as indexed by LC3-II levels. Rather, the benefits appeared to stem from intricate shifts in selective autophagy components, shedding light on the complexity of CR’s influence on cellular pathways. This nuanced interplay between nutrient sensing, autophagy regulation, and tumor suppression marks a significant advance in our understanding of cancer biology.
Experts suggest that the study’s findings may have profound implications for developing dietary or pharmacological strategies that mimic calorie restriction’s effects, potentially offering safer and more controlled approaches for cancer prevention and therapy, especially in individuals at high risk due to radiation exposure or other carcinogenic insults.
Moreover, this research highlights the importance of p62 as a molecular target for further investigation, with the potential to uncover novel therapeutics that modulate its expression or function to enhance the body’s natural defense mechanisms against tumor development.
While calorie restriction is impractical for widespread clinical application due to adherence challenges and potential side effects, the molecular pathways identified here open avenues for new interventions that harness the benefits of CR without requiring drastic dietary changes, ultimately transforming cancer prevention paradigms.
This study also contributes to a growing body of literature that questions the universality of increased autophagy as a cancer-suppressive mechanism, emphasizing instead the context-dependent roles of autophagy-related proteins. By parsing out these details, researchers aim to fine-tune strategies that maximize therapeutic efficacy while minimizing unintended consequences.
Beyond cancer, these findings on p62 and hepatic responses to nutritional and radiation stress echo broader themes in aging research, where caloric intake and cellular quality control emerge as pivotal factors influencing longevity and tissue resilience.
The availability of detailed mechanistic data from this study encourages interdisciplinary collaboration, combining molecular biology, nutrition science, radiation research, and clinical oncology to translate bench-side discoveries into effective bedside interventions.
Future research will likely focus on elucidating the signaling pathways downstream of p62 upregulation in calorie-restricted, irradiated organisms, potentially unraveling interactions with apoptosis, necrosis, or immune-mediated clearance mechanisms, thereby expanding our grasp of tumor microenvironment dynamics.
In sum, this seminal work uncovers a sophisticated molecular portrait of how calorie restriction maneuvers cellular processes to suppress radiation-induced liver carcinogenesis. It reveals that p62, rather than classical autophagy markers like LC3, plays a pivotal role in this cancer suppression, laying a foundation for innovative cancer prevention approaches grounded in molecular nutrition and cellular quality control.
Subject of Research: The study investigates the effects of calorie restriction on autophagy-related proteins and liver tumor development in radiation-exposed mice.
Article Title: Calorie restriction in radiation-exposed mice affects the expression of autophagy-related protein p62.
Article References:
Nakayama, T., Suzuki, K. & Mitsutake, N. Calorie restriction in radiation-exposed mice affects the expression of autophagy-related protein p62. BMC Cancer 25, 1388 (2025). https://doi.org/10.1186/s12885-025-14771-z
Image Credits: Scienmag.com