Water’s journey from a liquid state to a solid crystalline form is a phenomenon so familiar yet profoundly complex that it continues to baffle scientists at the molecular level. Despite its ubiquity in nature—manifesting as snow, hail, frost, and ice—the microscopic details governing how water transitions to ice, particularly at interfaces, remain enigmatic. Researchers at the Institute of Industrial Science, University of Tokyo, have now brought new clarity to this elusive process by examining the nuanced molecular interactions driving ice nucleation near surfaces using advanced molecular dynamics simulations. Their findings reveal a surprising mechanism that reshapes our understanding of how ice forms and propagates.
Fundamental knowledge dictates that water freezes at 0°C (32°F), but the actual phase transition from liquid to solid does not occur instantaneously upon reaching this threshold. Instead, freezing is initiated through nucleation, wherein tiny clusters of ice—nuclei—form and subsequently grow to solidify the bulk liquid. This nucleation phase is highly sensitive to ambient conditions such as temperature, but as the University of Tokyo researchers highlight, the microscopic environment close to material surfaces plays an even more decisive role. Understanding the molecular arrangement of water adjacent to surfaces is essential to unlocking the secrets behind why ice commonly initiates on surfaces before extending into the surrounding water volume.
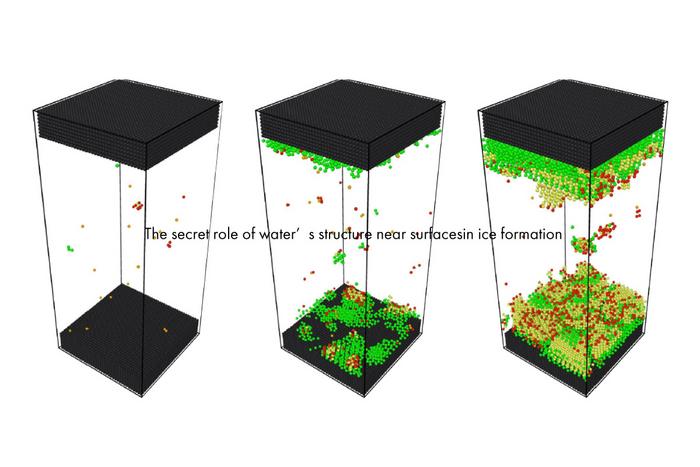
The lead author, Gang Sun, reflects on everyday observations: when a glass of water freezes, ice first materializes at the glass-water interface before gradually advancing inward. This phenomenon signals that the interface between water and another material fundamentally alters nucleation kinetics. Such interfacial effects are notoriously difficult to probe experimentally at the molecular scale, prompting the researchers to leverage molecular dynamics simulations. These simulations offer a window into the molecular choreography of water molecules near diverse surfaces, enabling precise quantification of physical parameters including temperature, molecular interaction strengths, and hydration effects.
Intriguingly, the simulations challenge conventional wisdom that suggested the surface’s ice affinity—its propensity to attract and stabilize ice crystals—directly controls nucleation pathways. Instead, the study uncovers that the microscopic structuring of water layers immediately adjacent to the surface exerts a defining influence on ice formation. Specifically, two water monolayers near the interface organize into a distinct bilayered hexagonal lattice that acts as a scaffold promoting nucleation. This low-dimensional crystalline architecture templates ice growth, effectively lowering the energetic barriers to nucleation and catalyzing freezing. Such precise molecular ordering near surfaces had remained elusive until now and reveals a fundamentally new conceptual framework for ice nucleation.
However, the story is nuanced. The team found that the hydrophilicity of the surface—its strength of attraction to water molecules—needs to strike an optimal balance. Excessive hydrophilicity disorders the critical bilayered hexagonal arrangement by overly anchoring water molecules, thereby disrupting the delicate lattice formation required for nucleation. Conversely, if the surface pulls too weakly on water molecules, the bilayer fails to stabilize, and nucleation is impeded. This forms a “Goldilocks zone” for surface interaction strength, where nucleation is most favorable, neither excessively strong nor weak. Such a paradigm complements existing theories but reframes how surfaces can be engineered to modulate freezing behavior precisely.
The implications of these insights extend far beyond fundamental physical chemistry into practical domains where controlling ice formation is critical. Designing anti-icing coatings based on these principles could revolutionize industries from aviation to renewable energy, where ice accumulation severely hampers performance and safety. Surfaces engineered to manipulate the bilayer hexagonal water structuring could either promote or inhibit ice nucleation on demand, offering unprecedented control at microscopic scales previously inaccessible.
Additionally, the researchers suggest that similar mechanisms may govern nucleation phenomena in other tetrahedrally bonded liquids beyond water. Substances like silicon and carbon, which exhibit analogous bonding geometries, might exhibit comparable nucleation dynamics at interfaces. This cross-material relevance opens new avenues in semiconductor manufacturing and materials science where controlling nucleation and crystallization is vital for producing high-quality materials with desired physical properties.
From a climate science perspective, improved understanding of ice nucleation at interfaces impacts models of atmospheric processes involving cloud formation and precipitation. Nucleation often dictates cloud microphysics and thus influences weather patterns and climate regulation. The new insights into molecular structuring may help refine climate models and guide geoengineering strategies to mitigate adverse environmental effects.
Methodologically, this breakthrough underscores the power of molecular dynamics simulations combined with rigorous physical parameter exploration. The team systematically varied temperature and intermolecular forces to isolate the impact of molecular layering adjacent to surfaces, a feat unachievable by purely experimental means. This integration of theoretical and computational chemistry exemplifies the frontier of nanoscale science, where atomistic insights translate into macroscopic phenomena with wide-ranging implications.
The University of Tokyo team’s findings not only deepen our fundamental understanding of water’s polymorphic behavior but also pave the way for smart materials design aimed at harnessing or mitigating ice formation. Their submission, published on June 4, 2025, in the Journal of Colloid and Interface Science, stands as a landmark contribution to physical chemistry and surface science, marking a significant advance in the centuries-long quest to unravel water’s mysteries.
As studies on nucleation continue, this work highlights the critical importance of molecular-level phenomena that occur at interfaces—not only reshaping academic perspectives but potentially generating transformative applications across science and technology sectors. By illuminating the “secret role” of water’s structured layers near surfaces, the research invites a new era of innovation in manipulating phase transitions with precision and purpose.
Subject of Research: Molecular mechanisms of ice nucleation and the role of water structuring near surfaces.
Article Title: The secret role of water’s structure near surfaces in ice formation
News Publication Date: 4 June 2025
Web References: https://doi.org/10.1016/j.jcis.2025.137812
Image Credits: Institute of Industrial Science, The University of Tokyo
Keywords
Physical chemistry, Surface science, Wetting, Water chemistry, Ice, Nucleation, Water molecules, Surface tension, Crystal structure, Thermodynamics