Research into energy storage technologies is becoming increasingly crucial as the global demand for energy intensifies. Efficient and safe systems are paramount in promoting the continued growth of renewable energy resources. In the realm of rechargeable aqueous zinc metal batteries (AZMBs), considerable attention has emerged due to their intrinsic advantages, such as high safety levels, environmental sustainability, extensive availability of zinc resources, and favorable energy density characteristics. Despite their promise, commercial viability of AZMBs is hampered by substantial challenges, primarily the instability of the zinc anode, a formidable hurdle that requires urgent attention.
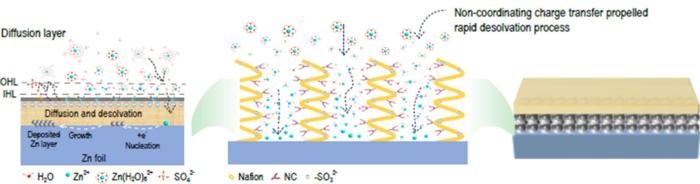
When embedded in aqueous solutions, zinc ions predominantly exist as hydrated complexes, particularly denoted as [Zn(H2O)6]2+. This phenomenon introduces several complications, notably when these hydrated ions approach the electrode interface. Here, water molecules interact directly with the zinc metal, precipitating unwanted chemical reactions such as hydrogen evolution reactions (HER), which in turn lead to corrosion and passivation. Such processes significantly contribute to the rapid degradation of the electrode, limiting the overall performance and lifespan of the battery. Furthermore, the slow kinetics of desolvation for Zn2+ ions leads to non-uniform deposition of zinc, fostering the growth of dendrites. These dendrites pose severe risks, as they can penetrate the separator, inciting short circuits that may threaten device safety.
In response to these considerable challenges, researchers from the Shenzhen Institutes of Advanced Technology at the Chinese Academy of Sciences have introduced an innovative design for an artificial solid electrolyte interface (SEI) that employs non-coordinating charge transfer. Central to this novel approach is a composite coating featuring nitrogen-doped amorphous carbon (NC) combined with perfluorosulfonic acid polymer, known as Nafion. This artificial SEI layer is specifically engineered to optimize the behavior of zinc ion transport while efficiently mitigating side reactions that can undermine battery integrity.
Nafion plays a crucial role in this advanced interface by serving as a selective ion channel. It strategically blocks anions and water molecules, thereby minimizing direct interaction with zinc metal and enhancing the stability of the system. Concurrently, the inclusion of NC material allows for the enhancement of the Fermi level within the structure, enabling a non-coordinating charge transfer mechanism. This synergistic approach effectively facilitates the desolvation process of Zn2+ ions and promotes a more uniform deposition of zinc, which is essential for enhancing battery performance.
Experimental validations indicate that the application of this artificial SEI leads to significant improvements in the stability of AZMBs. The NC-Nafion@Zn symmetric battery, featuring this advanced design, has demonstrated an impressive operational lifespan of 3400 hours at a current density of 1 mA cm-2 and 2000 hours at 5 mA cm-2. Such figures are indicative of a substantial extension to the anode’s durability, including the capability to withstand higher operational loads without compromising performance.
Additionally, the full battery configuration—NC-Nafion@Zn paired with a manganese oxide and carbon nanotube (CNTs) mixture—exhibits exceptional cycling stability. This configuration has achieved a staggering 9300 cycles at a current density of 2 A g-1 while retaining 91.3% of its capacity over the cycling period. Such performance levels are markedly superior to those observed in existing technologies, indicating a significant leap forward in battery functionality.
Moreover, the implementation of the artificial SEI demonstrates a remarkable ability to suppress hydrogen evolution reactions. This remarkable capacity leads to an enhanced coulombic efficiency (CE) measured at 99.1%, which speaks to the improved charge-discharge reversibility and overall energy utilization prowess of the battery system. Such developments not only bolster the immediate potential of AZMBs but also inspire broader applications across energy-storage solutions.
To explore the practical implications and feasibility of their findings, the research team undertook a series of tests in pouch cells, successfully powering an LED array. This demonstration validates the practical application potential of the developed technology, establishing its relevance for large-scale energy storage solutions, grid frequency regulation, and portable power devices. The versatility of the NC-Nafion composite approach underscores its promising market prospects.
In conclusion, this study introduces a groundbreaking design for the artificial solid electrolyte interface, effectively addressing the persistent challenges related to zinc anode stability. By leveraging the principles of Fermi-level modulation and non-coordinating charge transfer, researchers have successfully enhanced both the cycle life and safety of rechargeable aqueous zinc metal batteries. The innovative nature of this approach not only aims toward immediate performance enhancement but also sets the groundwork for future advancements in energy storage systems, marking a pivotal moment in rechargeable battery technology.
Subject of Research: Development of an artificial solid electrolyte interface for aqueous zinc metal batteries
Article Title: Innovative Design of Artificial SEI Enhances Zinc Anode Stability in Aqueous Zinc Metal Batteries
News Publication Date: October 2023
Web References: http://dx.doi.org/10.1093/nsr/nwaf070
References: National Science Review
Image Credits: ©Science China Press
Keywords: Aqueous zinc metal batteries, artificial solid electrolyte interface, zinc anode stability, energy storage, rechargeable batteries, electrochemistry.