A groundbreaking advancement in molecular imaging now offers hope for faster and more precise evaluation of treatment responses in rheumatoid arthritis (RA) patients. Traditionally, assessing the effectiveness of anti-tumor necrosis factor (aTNF) therapies has required a waiting period of three to six months to confirm clinical improvement. However, recent research published in The Journal of Nuclear Medicine details a novel PET/CT imaging tracer that can detect therapeutic response within just four weeks, potentially revolutionizing personalized treatment pathways in RA care.
Rheumatoid arthritis is a chronic autoimmune condition marked by persistent inflammation of the synovial joints, leading to gradual degradation of cartilage and bone. This progressive damage can severely impair mobility and quality of life if not halted or slowed by effective intervention. The heterogeneity of patient responses to aTNF therapy—effective in only about half to two-thirds of cases—further complicates disease management. This makes early and accurate biomarkers essential to streamline therapy selection and prevent unnecessary side effects or lost time on ineffective treatments.
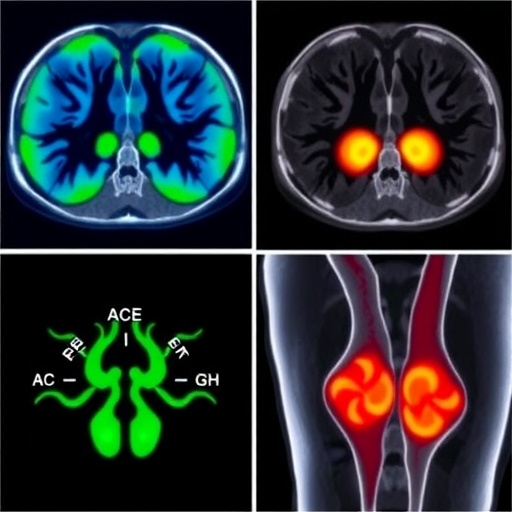
Central to the pathophysiology of RA are macrophages, a type of immune cell deeply involved in initiating and perpetuating synovial inflammation. Their abundance and activation correlate strongly with disease severity and progression. Recognizing this, researchers led by Wouter van Binsbergen at the Amsterdam University Medical Center have employed a cutting-edge PET/CT tracer, [^11C]DPA-713, which selectively binds to macrophage-associated translocator proteins within inflamed joint tissues. This allows for direct visualization and quantification of macrophage burden and activity—a crucial innovation in molecular imaging for inflammatory arthritis.
In a controlled study involving 20 RA patients beginning aTNF treatment, [^11C]DPA-713 PET/CT scans were performed at baseline and again after four weeks. Analysis focused on standardized uptake values (SUVs), a quantitative measure of tracer accumulation across 44 individual joints. These SUV metrics were then correlated with clinical disease activity scores assessed after 26 weeks of therapy, providing an early indicator of treatment success or failure.
The findings were striking: early PET/CT measurements exhibited a statistically significant association with long-term clinical outcomes. Elevated tracer uptake in specific joint clusters corresponded to persistent disease activity, while reductions at four weeks portended improved clinical responses months later. Importantly, combining PET/CT data with conventional clinical assessments enhanced predictive accuracy, underscoring the potential of integrated diagnostic approaches in rheumatology.
This non-invasive imaging modality holds profound implications for personalized medicine. By reliably identifying responders and non-responders shortly after treatment initiation, clinicians can tailor therapeutic regimens more effectively, sparing patients from ineffective drugs and associated toxicities. Furthermore, the ability to detect macrophage dynamics as a biomarker may facilitate the development of novel therapeutics targeting distinct immune pathways underlying RA.
Beyond rheumatoid arthritis, the study’s success heralds broader applications of immune cell-targeted PET/CT imaging across other inflammatory and autoimmune disorders. Harnessing such molecular imaging technologies can transform patient stratification, treatment monitoring, and drug development pipelines by providing actionable biological insights rather than relying solely on symptomatic evaluation.
Dr. van Binsbergen emphasized that these findings advocate for wider adoption of molecular imaging in clinical practice and research: “Our study demonstrates that macrophage-specific PET tracers like [^11C]DPA-713 can offer early, objective evaluation of therapeutic response. This represents a significant leap forward in managing RA and exemplifies the promise of precision imaging techniques for a range of immune-mediated diseases.”
The methodology involved meticulous whole-body imaging protocols using state-of-the-art PET/CT scanners capable of capturing detailed metabolic activity within small joints prone to RA pathology, such as those in the hands. Sophisticated image analysis, including standardized uptake quantification and regional joint clustering, enabled robust correlations with established clinical metrics.
While the sample size was relatively modest, the strong statistical associations and the biologically plausible mechanism of macrophage targeting provide a compelling rationale for further large-scale validation studies. Future research will also explore optimizing tracer dosing, scanning intervals, and integration with other biomarkers to maximize predictive performance.
This research not only promises to expedite decision-making in RA management but also exemplifies the convergence of immunology, nuclear medicine, and bioengineering. By visualizing immune cells in vivo, it allows a window into previously occult disease processes and fosters a more dynamic understanding of treatment effects, potentially enabling adaptive therapeutic strategies.
For clinicians and patients alike, this innovation underscores a transformative shift from reactive to proactive care. It empowers earlier intervention adjustments based on real-time biological data, aligning treatments more closely with individual disease trajectories. As molecular imaging tools continue to evolve, their incorporation into routine rheumatology practice could fundamentally improve outcomes and quality of life for millions affected by rheumatoid arthritis worldwide.
Subject of Research: Early therapeutic evaluation of anti-tumor necrosis factor treatment in rheumatoid arthritis using macrophage-targeted PET/CT imaging.
Article Title: Macrophage-Targeted [11C]DPA-713 PET/CT Imaging for Early Therapeutic Evaluation of Anti–Tumor Necrosis Factor Treatment in Rheumatoid Arthritis
News Publication Date: February 27, 2026
Web References: https://doi.org/10.2967/jnumed.125.270289
References: van Binsbergen WHJ, de Jongh J, Voskuyl AE, van der Laken CJ, Yaqub M, Zwezerijnen GJC, van der Pas S, van Schaardenburg D. Macrophage-Targeted [^11C]DPA-713 PET/CT Imaging for Early Therapeutic Evaluation of Anti-Tumor Necrosis Factor Treatment in Rheumatoid Arthritis. The Journal of Nuclear Medicine. 2026.
Image Credits: W. van Binsbergen, MSc, MD, PhD-student, Amsterdam University Medical Center, Amsterdam, The Netherlands.
Keywords: Molecular imaging, Rheumatoid arthritis, Positron emission tomography, Macrophage targeting, [^11C]DPA-713, Anti-TNF therapy, PET/CT imaging, Immune cell imaging, Early therapeutic evaluation, Personalized medicine.